Taking Stock of CAR T-Cell Therapy
Five years after the FDA approved the first CAR T-cell therapy, use of the treatment has expanded to many patients with blood cancer.


Five years after the FDA approved the first CAR T-cell therapy, use of the treatment has expanded to many patients with blood cancer.
Researchers aim to minimize disease relapses by targeting multiple proteins.

Aggressive treatments given to younger patients with B-cell non-Hodgkin lymphoma may leave them vulnerable to other diseases later in life, according to a study in the AACR journal Cancer Epidemiology, Biomarkers & Prevention.
The FDA approved zanubrutinib, a tyrosine kinase inhibitor, for this form of non-Hodgkin lymphoma. The U.S. Food and Drug Administration (FDA) approved zanubrutinib (Brukinsa) for adult patients with Waldenström macroglobulinemia, a slow-growing form of...
The FDA approved a CAR T-cell therapy – a class of cancer immunotherapy – to treat certain patients with large B-cell lymphoma, the most common form of non-Hodgkin lymphoma.
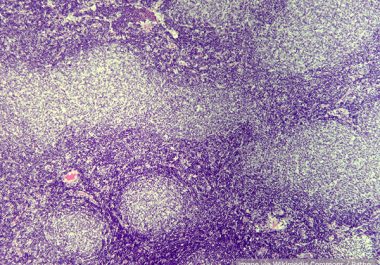
The FDA granted accelerated approval to a molecularly targeted therapeutic for certain patients with marginal zone lymphoma or follicular lymphoma.
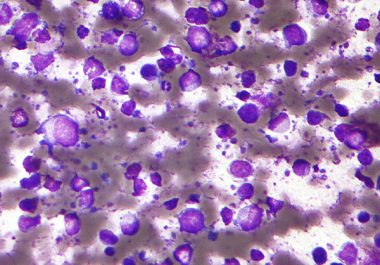
The FDA has approved a new immunotherapeutic for treating certain adult patients with diffuse large B-cell lymphoma. The U.S. Food and Drug Administration (FDA) granted accelerated approval to a new targeted immunotherapeutic called tafasitamab-cxix...
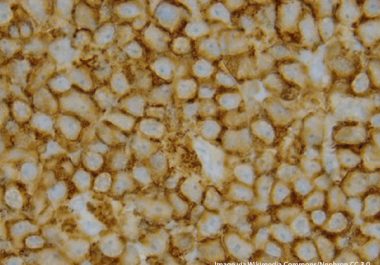
The FDA has approved a CAR T-cell immunotherapy to treat certain adult patients with mantle cell lymphoma.
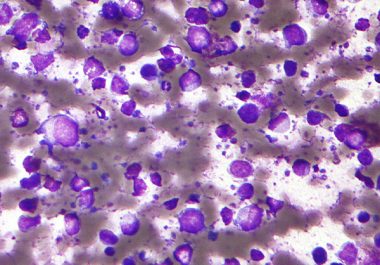
The FDA has approved a targeted therapy drug to treat certain adult patients with diffuse large B-cell lymphoma.
The FDA has approved an alternative dosing schedule for the immune checkpoint inhibitor pembrolizumab.