Advancing Biomarker Testing for Four Common Cancers
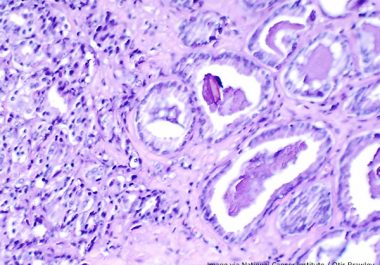
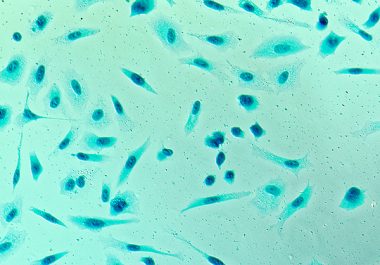
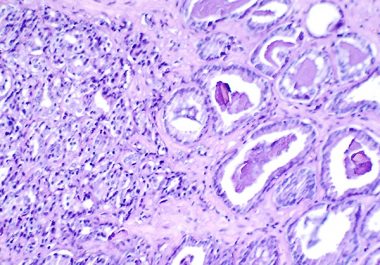
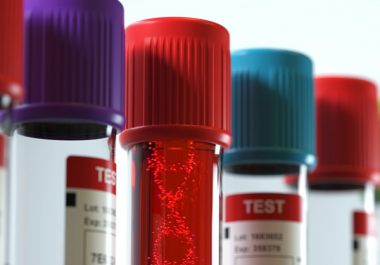
The FDA granted approval to expand the use of a minimally invasive biomarker test to identify certain patients with ovarian, lung, breast, or prostate cancer eligible for treatment with specific molecularly targeted therapeutics.