Gauging Progress Against Cervical Cancer
January is Cervical Cancer Awareness Month. Given that January was named after the Roman god Janus, whose two faces allowed him to look both backward into the old year and forward into the new one, it seemed a good time to look back at the progress we made against cervical cancer in 2018 and to look for ways to build on the progress and further reduce the incidence and mortality of the disease in the future.
Updating screening recommendations, but we must do better at implementation
In the past few decades, most of the progress that has been made against cervical cancer has been in the areas of prevention and early detection.
One of the most important strides forward was the development of the Pap test. Its widespread use for cervical cancer screening is credited with driving down the U.S. cervical cancer incidence rate. The latest National Cancer Institute (NCI) data show that the U.S. cervical cancer incidence rate fell 50 percent from 1975 to 2015.

In August 2018, after reviewing new data, the USPSTF updated its cervical cancer screening recommendations to add screening every five years with HPV testing alone. The new recommendations provide women ages 30 to 65 at average risk for developing cervical cancer three options for screening: Pap testing alone every three years, HPV testing alone every five years, or co-testing every five years. Pap testing alone every three years remains the recommended cervical cancer screen for women ages 21 to 29 at average risk for developing the disease.
In adding a new cervical cancer screening option, the USPSTF hopes to increase the number of women who get screened. This is vital because research shows that most cases of cervical cancer occur among women who have not been adequately screened, meaning they have not been screened in the past five years or more.
The latest data show that the percentage of women ages 21 to 65 who were up to date with cervical cancer screening fell slightly from about 86 percent in 2000 to 83 percent in 2015. Screening rates in 2015 were particularly low among certain population groups, including Asian women (75.8 percent), American Indian/Alaska Native women (76.9 percent), and Hispanic women (78.6 percent), as well as women who were uninsured (63.8 percent), had not completed high school (71.2 percent), or had the lowest incomes (75.2 percent). Addressing disparities in screening is vitally important if we are to significantly reduce the incidence and mortality of cervical cancer. In fact, it is one of the areas identified as needing significant research by the USPSTF in its Eighth Annual Report to Congress on High-Priority Evidence Gaps for Clinical Preventive Services, which was released in November 2018.
Expanding HPV vaccine eligibility, but we need greater uptake
The discovery of the link between HPV and cervical cancer also led to the development and U.S. Food and Drug Administration (FDA) approval of three vaccines that can prevent infection with certain HPV strains. The first two vaccines approved by the FDA, in 2006 and 2009, protect against only the two most common cancer-causing strains of HPV—HPV16 and 18. Gardasil 9, the third vaccine approved by the agency, in 2014, protects against seven cancer-causing strains of HPV—HPV16, 18, 31, 33, 45, 52, and 58. As of 2017, only Gardasil 9 is available in the United States.
In October 2018, the FDA approved expanding the use of Gardasil 9 to include women and men ages 27 to 45. Before then, it was approved only for vaccination of females and males ages 9 to 26. In announcing the decision, Peter Marks, MD, PhD, director of the FDA’s Center for Biologics Evaluation and Research, said that the approval represents an important opportunity to help prevent HPV-related diseases and cancers in a broader age range.
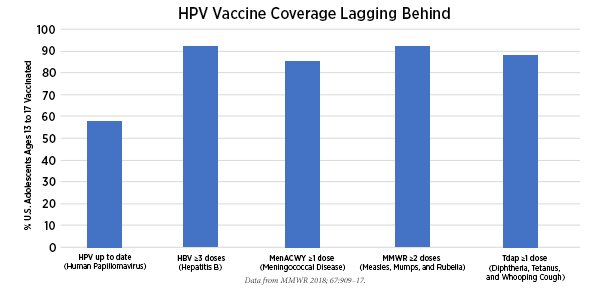
Despite the knowledge that Gardasil 9 can prevent infection with the seven strains of HPV responsible for about 90 percent of invasive cervical cancers, the latest data show that just 49 percent of adolescents ages 13 to 17 were up to date with HPV vaccination in 2017. The current Centers for Disease Control and Prevention recommendation is that all children age 11 or 12 should get two shots of HPV vaccine six to 12 months apart and that anyone being vaccinated after the age of 14 should have three shots over the course of six months.
This low HPV vaccine coverage contrasts starkly with coverage for other childhood vaccines. For example, 92 percent of adolescents ages 13 to 17 have received two or more doses of the MMR vaccine that protects against measles, mumps, and rubella.
In June 2018, the American Association for Cancer Research (AACR) joined 70 NCI-designated cancer centers and five national cancer organizations in urging parents, young adults, and physicians to act to increase HPV vaccination rates and screening access to eliminate HPV-related cancers, including cervical cancer.
Increasing treatment options, but we need more
Despite the dramatic decrease in the cervical cancer incidence rate from 1975 to 2015, NCI data show that there was a slight reduction in the overall five-year relative survival rate for women diagnosed with the disease over the same period. In 1975, it was 69 percent, whereas the most recent data put this number at just over 66 percent. For women diagnosed with metastatic cervical cancer the prognosis is even worse; the five-year relative survival rate for these patients is just 17 percent.
The good news is that in 2018, there were several advances in cervical cancer treatment.
As discussed in a previous post on this blog, in June 2015, the FDA approved the immunotherapeutic pembrolizumab (Keytruda) for treating patients with recurrent or metastatic cervical cancer that tests positive for the protein PD-L1 and that has progressed despite treatment with cytotoxic chemotherapy.
In October 2018, two papers published in The New England Journal of Medicine provided new insight into different options for surgery for early-stage cervical cancer. The National Comprehensive Cancer Network (NCCN) recommends that women with early-stage cervical cancer (stages 1A2 to 2A) have a radical hysterectomy, which means they have surgery to remove the uterus, cervix, and part of the vagina; and sometimes the ovaries, fallopian tubes, and nearby lymph nodes. This surgery can be done as open surgery (laparotomy), whereby the surgeon makes one large cut through the abdominal wall to perform the surgery, or as minimally invasive laparoscopic surgery, where the surgeon makes small incisions and performs surgery aided by a camera.
Since the first report of laparoscopic surgery for early-stage cervical cancer in 1992, minimally invasive surgery has become widely used because it is typically associated with shorter hospital stays and lower risk of complications after surgery. Despite this, there is no high-quality evidence from randomized trials investigating whether there are any survival differences following the two different types of radical hysterectomy, as highlighted by the authors of both The New England Journal of Medicine papers.
The authors of the first paper conducted a randomized trial to compare survival for women with early-stage cervical cancer who had open surgery (312 women) or minimally invasive surgery (319 women) for their radical hysterectomy. They found that minimally invasive surgery was associated with lower rates of disease-free survival and overall survival than open surgery. Three-year rates of disease-free and overall survival were 91.2 percent and 93.8 percent, respectively, for the minimally invasive surgery arm, and 97.1 percent and 99.0 percent for the open surgery arm.
In the second paper, the authors report a retrospective analysis of outcomes of 2,461 women who had a radical hysterectomy after an early-stage cervical cancer diagnosis; 49.8 percent had minimally invasive surgery and 51.2 percent had open surgery. They found that minimally invasive surgery was associated with shorter overall survival than open surgery; the four-year mortality was 9.1 percent among women who had minimally invasive surgery and 5.3 percent among those who had open surgery.
After some of these data were presented early in 2018 at the Society of Gynecologic Oncology annual meeting in New Orleans, the NCCN updated its cervical cancer guidelines to include a statement recommending that women be carefully counseled about the risks and benefits of open surgery and minimally invasive surgery to ensure that they make the most informed decision about their care.
Despite the progress, we must do more. More than 4,000 U.S. women are predicted to have died from the disease in 2018, making it the 20th most common cause of death from cancer in the United States. Worldwide, cervical cancer has an even greater toll. It is the ninth-leading cause of death from cancer, with more than 300,000 women predicted to have died from the disease in 2018. We need more research to identify new approaches to implementing our current, effective tools for cervical cancer prevention and screening, and new ways to treat the disease. We also need to continue efforts to increase public awareness about the availability and effectiveness of vaccines and cervical cancer screening, which can prevent cervical cancer.