Editors’ Picks, March 2026: Pediatric Leukemia Development, Low-dose Tamoxifen Use, and More
As we welcome the greening season, check out this month’s Editors’ Picks, which include new insights into how developmental state and molecular plasticity of target cells, together with genetic lesions, drive development of pediatric leukemia; challenges in improving awareness of the role of alcohol as a carcinogen; a study on higher uptake of low-dose tamoxifen for breast cancer chemoprevention; clinical trials in pancreatic and breast cancer; and more.
Read the abstracts of the studies highlighted by the editors of the 10 peer-reviewed journals of the American Association for Cancer Research (AACR) and follow the links to the full-text articles that will be freely available for a limited time.
Journal: Blood Cancer Discovery
Persistent fetal gene expression in childhood neoplasms is usually explained by a maturation block originating in the prenatal phase. In contrast, reactivation of fetal genes in adult malignancies is considered a consequence of oncofetal reprogramming (OFR) and is associated with aggressive disease. By reconstructing epigenetic ontogeny in juvenile myelomonocytic leukemia (JMML), we identified a postnatal maturation state of JMML stem cells with high transcriptional plasticity indicative of OFR in high-risk disease. Similarly, postnatal activation of oncogenic signaling by inducible Ptpn11E76K mutation in mice triggered molecular plasticity and reactivation of fetal gene expression. Integrative multi-omics analysis revealed aberrant CD52 expression as a feature of high-risk JMML stem cells. Anti-CD52 treatment depleted JMML stem cells and blocked disease propagation in xenograft models. Our results challenge the prevailing maturation block model of pediatric leukemogenesis and establish RAS-associated stem cell plasticity as a determinant of OFR and potential therapeutic vulnerabilities in high-risk JMML.
Significance: Persistent fetal gene expression in pediatric malignancies is considered a consequence of prenatal maturation blockade. In this study, we demonstrate that oncogenic PTPN11 mutations enhance cellular plasticity. This leads to partial restoration of fetal molecular programs, creating new therapeutically exploitable vulnerabilities.
A related commentary was published in the March issue, where this article was highlighted.
Journal: Cancer Discovery
Ontogeny Dictates Oncogenic Potential, Lineage Hierarchy, and Therapy Response in Pediatric Leukemia

Accumulating evidence links pediatric cancers to prenatal transformation events, yet the influence of the developmental stage on oncogenesis remains elusive. We investigated how hematopoietic stem cell developmental stages affect leukemic transformation, disease progression, and therapy response using a novel, humanized model of NUP98::NSD1-driven pediatric acute myeloid leukemia that is particularly aggressive with WT1 comutations. Fetal-derived hematopoietic stem cells readily transform into leukemia, and WT1 mutations further enhance stemness and alter lineage hierarchy. In contrast, stem cells from later developmental stages become progressively resistant to transformation. Single-cell analyses revealed that fetal-origin leukemia stem cells exhibit greater quiescence and reliance on oxidative phosphorylation than their postnatal counterparts. These differences drive distinct therapeutic responses despite identical oncogenic mutations. In patients, onco-fetal transcriptional programs correlate with worse outcomes. By targeting key vulnerabilities of fetal-origin leukemia cells, we identified combination therapies that significantly reduce aggressiveness, highlighting the critical role of ontogeny in pediatric cancer treatment.
Significance: This study signifies the critical consequences of developmental timing in cancer initiation, revealing that identical driver mutations in fetal- versus postnatal-origin leukemias exhibit fundamentally distinct biology and treatment responses. Recognizing these developmental differences opens avenues for personalized therapeutic strategies, improving outcomes for pediatric patients with aggressive disease subtypes in leukemia.
This article was featured on the cover of the March issue and in a commentary published in the March issue of Blood Cancer Discovery.
Journal: Cancer Epidemiology, Biomarkers & Prevention
Background: Alcohol use increases the risk of seven cancers, including breast and colorectal cancers. Awareness of this is low in the US population (34%), which impedes risk reduction efforts, support for alcohol regulation policies, and physician and family communication about this topic. Efforts to improve awareness of the cancer risks of alcohol will require multiple approaches, including the use of educational messages tailored to audience needs. There is a lack of evidence about how existing messaging influences awareness and characteristics associated with awareness that could guide future messaging.
Methods: We recruited a nationally representative sample of N = 827 US adults aged ≥21 who reported alcohol use in the past month and were unaware that alcohol increases cancer risk. Participants viewed a publicly available educational video on this topic and subsequently reported cancer risk awareness.
Results: After video exposure, 70% of participants reported awareness. Post-video awareness was lower among heavier drinkers, those with a cancer history, those with no family cancer history, and those who tend to avoid health information, perceive low cancer controllability, or feel cancer information overload. Those who reported inaccurate beliefs at baseline (responding “no” to the question of whether cancer can result from too much alcohol) reported lower awareness than those who initially reported that they “didn’t know” whether alcohol increased cancer risk.
Conclusions: These findings provide groundwork to guide the development of adaptations to available alcohol and cancer risk messaging.
Impact: Educational messages tailored to audience needs can help inform all sectors of the US public about the cancer harms of alcohol.
A related commentary was published in the March issue.
Journal: Cancer Immunology Research

Pancreatic ductal adenocarcinoma (PDAC) remains highly lethal and has poor immunogenicity, warranting the use of vaccines to guide and recruit the immune response. Building on prior efforts to achieve clinical immunotherapeutic responses against PDAC, we conducted a phase II study (NCT03190265) that combined attenuated mesothelin (MSLN)-secreting listeria vaccine (CRS-207) and GM-CSF–secreting allogeneic whole-cell vaccine (GVAX) along with checkpoint inhibition. Patients with metastatic PDAC who progressed on chemotherapy were enrolled in one of two treatment arms in a randomized fashion. CRS-207 was given with anti–PD-1 (nivolumab) and anti-CTLA4 (ipilimumab) with (arm A) or without (arm B) GVAX. The primary endpoint was the objective response rate (ORR), and the secondary endpoint was safety. Fifty-seven patients received at least one dose of treatment, with two partial responses (4% ORR), in both arm B. The response rates were not significantly different between the two arms. Related grade ≥3 adverse events were seen in 39 (68%) patients, including 33 events attributed to CRS-207. Mass cytometry analysis of serially obtained biospecimens demonstrated treatment-induced promotion of T-cell memory and infiltration into the tumor microenvironment (TME). Peptide-specific T-cell expansions in vitro, followed by T-cell receptor sequencing, revealed clones specific to MSLN and mutant KRAS within the tumor. Accompanying these antitumor T-cell responses was significant enrichment of myeloid cells. High myeloid and regulatory T-cell signatures correlated with poor responses. We conclude that GVAX/CRS-207 plus nivolumab and ipilimumab successfully generates and expands T-cell clones specific to MSLN and mutant KRAS within the PDAC TME, but immunotherapy-induced myeloid cell enrichment remains a barrier to greater efficacy.
This article was featured on the cover of the March issue.
Journal: Cancer Prevention Research
In 2019, professional guidelines incorporated low-dose tamoxifen as an option for breast cancer chemoprevention among women with atypical hyperplasia (AH) or lobular or ductal carcinoma in situ (LCIS/DCIS). We assessed factors associated with low-dose tamoxifen use among women diagnosed with AH, LCIS, or DCIS from 2016 to 2019 and from 2020 to 2023 compared with full-dose selective estrogen receptor modulators (SERM) or aromatase inhibitors (AI) at Columbia University Irving Medical Center in New York City. Uni- and multivariable logistic regression were used to calculate odds ratios (OR) and 95% confidence intervals (CI) for variables associated with low-dose tamoxifen use. Among 2,260 evaluable women, 834 (36.9%) initiated a SERM or AI, and 140 (6.2%) took low-dose tamoxifen. Comparing women diagnosed before or after 2019, chemoprevention uptake significantly increased (33.9% vs. 39.3%, P = 0.008), particularly low-dose tamoxifen (3.3% vs. 8.6%). Among women who initiated chemoprevention, diagnosis of high-risk breast lesions before age 50 (OR = 3.02; 95% CI, 1.99–4.58), diagnosis after 2019 (OR = 2.83; 95% CI, 1.81–4.41), AH/LCIS versus DCIS (OR = 2.90; 95% CI, 1.95–4.31), and medical oncology referral (OR = 1.61; 95% CI, 1.02–2.54) were significant predictors of low-dose tamoxifen use. Those who initiated low-dose tamoxifen as their first chemoprevention had the lowest 1-year discontinuation rate (24.3%) compared with full-dose SERMs/AIs (32.3%–37.9%, P = 0.027). Since 2019, we observed a significant increase in low-dose tamoxifen use and chemoprevention uptake overall. Among women who initiated chemoprevention, low-dose tamoxifen uptake was higher among younger women and those with less advanced breast lesions. Low-dose options of proven chemopreventive agents may increase acceptance of risk-reducing medications for breast cancer prevention.
Prevention Relevance: This study provides evidence of increased use of low-dose tamoxifen in a prevention setting, particularly among younger women and those with less advanced breast lesions. Further research is needed to understand how to incorporate low-dose tamoxifen into patient–provider discussions to improve chemoprevention decision-making.
Journal: Cancer Research (March 1 issue)
Androgen Receptor Drives Polyamine Synthesis, Creating a Vulnerability for Prostate Cancer
Supraphysiologic androgen (SPA) treatment can paradoxically restrict the growth of castration-resistant prostate cancer (CRPC) with high androgen receptor (AR) activity, which is the basis for the use of bipolar androgen therapy (BAT) for patients with this disease. Although androgens are widely appreciated for enhancing anabolic metabolism, how SPA-mediated metabolic changes alter prostate cancer progression and therapy response is unknown. In this study, we report that SPA markedly increased intracellular and secreted polyamines in prostate cancer models. AR binding at enhancer sites upstream of the ornithine decarboxylase 1 (ODC1) promoter increased the abundance of ODC, a rate-limiting enzyme of polyamine synthesis, and de novo synthesis of polyamines from arginine. SPA-stimulated polyamines enhanced prostate cancer fitness, as dCas9-KRAB–mediated inhibition of AR regulation of ODC1 or direct ODC inhibition by difluoromethylornithine (DFMO) increased the efficacy of SPA. Mechanistically, AR activation combined with the loss of negative feedback by polyamines increased the activity of S-adenosylmethionine decarboxylase 1, leading to the depletion of its substrate, S-adenosylmethionine, and global protein methylation. These data provided the rationale for a clinical trial testing the safety and efficacy of BAT in combination with DFMO for patients with metastatic CRPC. Pharmacodynamic studies of this therapeutic combination in the first five patients in the trial indicated that this approach effectively depleted polyamines in plasma. Thus, the AR potently stimulates polyamine synthesis, which constitutes a vulnerability in prostate cancer treated with SPA that can be targeted therapeutically.
Significance: Increased polyamine synthesis is a prominent metabolic change induced by the androgen receptor that drives tumor progression and confers a targetable vulnerability in advanced prostate cancer.
A related commentary was published in the March 1 issue.
Journal: Cancer Research (March 15 issue)
Microglia Display Heterogeneous Initial Responses to Disseminated Tumor Cells
Brain metastases are frequent and often lethal complications of advanced cancers. Microglia, resident immune cells of the brain, are known to exert both anti- and protumor functions in late-stage metastases; however, their response during the initial outgrowth of metastatic lesions is not well characterized. Understanding how heterogeneous microglial subgroups are regulated in the developing tumor microenvironment could pave the way for therapeutic strategies to eliminate metastatic tumors at an early stage. In this study, we used a combination of in vivo fate map imaging, single-cell RNA sequencing, and a holographic photoconversion-based technique (opto-omics) to track tumor fate and early microglial responses over time in the same animals during the colonization of disseminated tumor cells. The microglial population was transcriptionally and morphologically heterogeneous, comprising both pro- and antitumor subsets. Genetic and pharmacologic perturbations revealed that microglial phenotypes could be shifted by inhibiting TGFβ signaling or by deleting the tumor cell surface antigens CD24 and CD47. These findings reveal targetable plasticity in early-stage microglial responses to brain metastasis and suggest that harnessing prophagocytic microglial states may offer a therapeutic window before systemic immunosuppression becomes dominant.
Significance: In vivo imaging with optical labeling and transcriptomics reveals heterogeneous microglia and identifies that CD24/CD47 loss or TGFβ modulation alters subpopulation fate, exposing a therapeutic window and actionable targets for brain metastases.
This article was featured on the cover of the March 15 issue, where a related commentary was also published.
Journal: Clinical Cancer Research (March 1 issue)
Purpose: Pembrolizumab plus olaparib versus pembrolizumab plus chemotherapy was evaluated as postinduction therapy for patients with PD-L1–unselected locally recurrent inoperable/metastatic triple-negative breast cancer (TNBC) who derived clinical benefit from first-line pembrolizumab plus platinum-based chemotherapy induction therapy.
Patients and Methods: In this phase 2 study (NCT04191135), participants with previously untreated locally recurrent inoperable/metastatic TNBC received first-line pembrolizumab 200 mg once every 3 weeks plus platinum-based chemotherapy. Participants with complete/partial response or stable disease (per RECIST v1.1) were randomly assigned 1:1 to pembrolizumab 200 mg once every 3 weeks plus olaparib 300 mg twice a day or pembrolizumab plus chemotherapy. Progression-free survival (PFS) and overall survival (OS) were primary endpoints.
Results: Of 460 participants receiving induction treatment, 271 were randomly assigned to postinduction therapy. The median PFS was 5.5 months in the pembrolizumab plus olaparib group versus 5.6 months in the pembrolizumab plus chemotherapy group [hazard ratio (HR), 0.98; 95% confidence interval (CI), 0.72–1.33; P = 0.4556]. The median OS was 25.1 versus 23.4 months, respectively (HR, 0.95; 95% CI, 0.64–1.40). In participants with tumor BRCA1/BRCA2 mutations (tBRCAm), HRs for PFS (HR, 0.70; 95% CI, 0.33–1.48) and OS (0.81; 95% CI, 0.28–2.37) favored pembrolizumab plus olaparib. Treatment-related adverse events occurred in 84.4% and 96.2% of participants receiving pembrolizumab plus olaparib and pembrolizumab plus chemotherapy, respectively.
Conclusions: Although the primary endpoint was not met, postinduction pembrolizumab plus olaparib therapy resulted in similar PFS and OS compared with pembrolizumab plus chemotherapy in this setting. The positive trend for PFS and OS in participants with tBRCAm suggests a potential nonchemotherapy strategy for maintaining clinical benefit attained with first-line pembrolizumab plus chemotherapy induction treatment. No new safety signals were identified.
Journal: Clinical Cancer Research (March 15 issue)
Purpose: Establish the safety, tolerability, and preliminary activity of trastuzumab deruxtecan (T-DXd) in combination with other anticancer therapies in human epidermal growth factor receptor 2 (HER2)–low metastatic breast cancer (mBC).
Patients and Methods: DESTINY-Breast08 was a two-part, open-label, multicenter, phase Ib study. Patients with locally confirmed HER2-low mBC received T-DXd plus capecitabine, durvalumab + paclitaxel, capivasertib, anastrozole, or fulvestrant. Eligibility criteria for hormone receptor status varied across modules and between study parts. Primary objectives were safety/tolerability and determining recommended phase II doses (RP2D); secondary endpoints included objective response rate (ORR; per investigator).
Results: In the dose-finding phase, 37 patients were assigned to a module. RP2Ds were determined for T-DXd plus capecitabine, capivasertib, anastrozole, or fulvestrant. For strategic reasons, T-DXd + durvalumab + paclitaxel was not pursued beyond the dose-finding phase (n = 3). In the dose-expansion phase, 101 patients were assigned to a module. For T-DXd + capecitabine, grade ≥3 adverse events (AE) occurred in 55% (11/20) of patients, and the ORR was 60%. For T-DXd + capivasertib, grade ≥3 AEs occurred in 67.5% (27/40) of patients, and the ORR was 60%. For T-DXd + anastrozole, grade ≥3 AEs occurred in 47.6% (10/21) of patients, and the ORR was 71.4%. For T-DXd + fulvestrant, grade ≥3 AEs occurred in 55% (11/20) of patients, and the ORR was 40%. Adjudicated drug-related interstitial lung disease/pneumonitis events were reported for T-DXd + capecitabine (3/20; grade 2, n = 2; grade 5, n = 1), T-DXd + capivasertib (8/40; all grade ≤2), and T-DXd + fulvestrant (5/20; all grade 2).
Conclusions: Safety results were generally consistent with known individual profiles for T-DXd and combination drugs. T-DXd plus capecitabine, capivasertib, anastrozole, or fulvestrant demonstrated preliminary clinical activity in patients with HER2-low mBC.
Journal: Molecular Cancer Research
BCOR Mutations Deregulate Cell Cycle and Hypoxic Adaptation Pathways in Retinoblastoma
Retinoblastoma is the most common pediatric eye cancer. Most cases of retinoblastoma are initiated by biallelic mutational inactivation of the RB1 gene, yet most retinoblastoma tumors harbor additional genomic aberrations that may promote tumor progression. After RB1, the gene that is most commonly mutated gene in retinoblastoma is BCOR, which is mutated in approximately 20% of retinoblastoma tumors and is associated with a more aggressive tumor phenotype and worse patient outcomes. Despite its importance, little is known about the role of BCOR in retinoblastoma. In this study, we interrogated BCOR in low-passage retinoblastoma cell lines using mass spectrometry, chromatin immunoprecipitation sequencing, and RNA sequencing. We show that the BCOR protein interacts with members of the noncanonical polycomb repressive complex 1.1 and localizes at gene loci with traditionally activating and repressing chromatin markers. Loss of BCOR downregulates the expression of genes associated with cell-cycle regulation and upregulates genes associated with hypoxic adaptation. We conclude that BCOR mutations slow cell proliferation and drive hypoxic adaptation in retinoblastoma via epigenetic mechanisms that may be amenable to targeted therapy.
Implications: This study reveals that BCOR may play a noncanonical, multifaceted role in retinoblastoma with implications in cell cycle, differentiation, and hypoxic adaptation, ultimately shedding insight into its molecular framework for future therapeutic strategies.
This article was highlighted in the March issue.
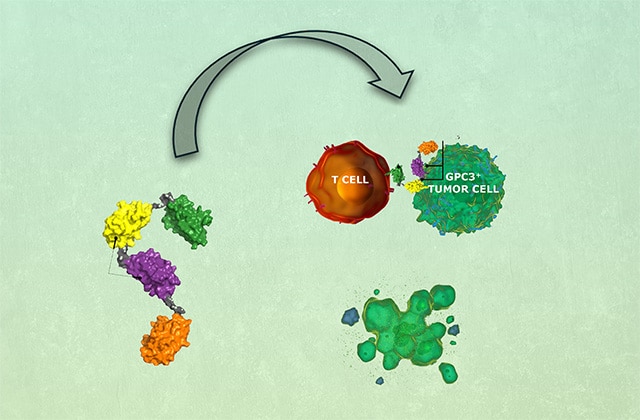
Journal: Molecular Cancer Therapeutics
T-cell engager (TCE) immunotherapy has demonstrated significant clinical activity in multiple cancers by inducing coengagement of T cells and tumor cells, resulting in T-cell activation and T cell–dependent cellular cytotoxicity (TDCC) against tumor cells. Current-generation TCEs are predominantly composed of antibody-based binding domains targeting the cluster of differentiation 3e molecule of the T-cell antigen receptor (TCR)–cluster of differentiation 3 complex on T cells and a tumor-associated antigen on tumor cells. However, limitations of this approach include cytokine release syndrome and a limited therapeutic window. In this study, we report the generation and preclinical evaluation of SAR444200, the first NANOBODY-based TCE clinical candidate binding to TCRαβ and glypican-3 (GPC3) to coengage T cells and GPC3+ tumor cells, causing TDCC. SAR444200 bound with nanomolar to picomolar affinity to TCRαβ and GPC3, respectively, and induced in vitro TDCC against multiple human tumor cell lines with differential GPC3 expression with picomolar potency. In vivo analysis using human cancer cell line–derived xenografts (HuH-7 and HepG2) in immunodeficient mice showed complete tumor regression at doses starting from 0.7 mg/kg. In exploratory non-human primate studies, intravenous administration of SAR444200 was well tolerated up to 8 mg/kg and exhibited greater than dose-proportional clearances and dose-proportional maximum concentrations across the tested dose range. The highly potent and efficacious activity of SAR444200 in diverse models of GPC3+ tumors and the extremely wide tolerated dose range merit further development of this compound. Furthermore, NANOBODY-based TCEs developed using an anti-TCRαβ moiety may have specific advantages for the development of TCEs.
This study was highlighted and featured on the cover of the March issue.
Journal: Cancer Research Communications
Epithelial ovarian cancer (EOC) represents the most lethal gynecologic malignancy, characterized by extensive tumor heterogeneity that contributes to treatment resistance and high recurrence rates. Recently, we developed SAIL66, a CLDN6-targeting T-cell engager currently in clinical evaluation for CLDN6-positive solid cancers, including EOC. Whereas CLDN6 is considered an attractive target for cancer therapy due to its cancer specificity, its biology remains poorly understood. In this study, we investigated the biological characteristics of CLDN6-positive EOC to identify its significance as a therapeutic target for ovarian cancer treatment. We demonstrated heterogeneous CLDN6 expression in xenograft and clinical tumors. In vitro–cultured ovarian cancer cell lines showed reversible changes in CLDN6 expression depending on cell density, accompanied by alterations in epithelial–mesenchymal transition (EMT)-related and stemness-related genes. Spatial transcriptomic analysis of clinical specimens revealed that CLDN6-positive areas formed both solid regions and dispersed small clusters within the same tumors, with differential expression of EMT-related and cell matrix remodeling genes between these areas, consistent with our in vitro observations at varying cell densities. Furthermore, carboplatin treatment increased CLDN6 expression, accompanied by changes in EMT-related genes. Leveraging these biological characteristics of CLDN6, we discovered that significant tumor regression was observed in mice treated with SAIL66 following carboplatin pretreatment. Post-carboplatin analysis revealed increased CLDN6 expression, EMT-related gene changes, and enhanced T-cell infiltration, which were associated with the synergistic effect of SAIL66. Our study provides insights into the biology and plasticity of CLDN6-positive cells in EOC heterogeneity and highlights the clinical significance of CLDN6-targeting therapies for ovarian cancer treatment.
Significance: CLDN6-positive ovarian cancer cells exhibit remarkable plasticity influenced by microenvironmental factors and chemotherapy, providing critical insights for understanding the biology of ovarian cancer progression and optimizing CLDN6-targeting therapy.



