From Bench to Bedside: How a Discovery About Menin Led to New Treatments for Acute Myeloid Leukemia
It all started with a picture.
This is the story of menin—starting with an X-ray crystal structure, which is a three-dimensional picture that captures the structure of a protein, and winding through numerous clinical trials, including AUGMENT‑101, KOMET‑001, KOMET-017, COVALENT-111, and COVALENT-112. A story written by many hands, funded across disciplines, presented in crowded poster halls, debated in plenaries, and engraved finally into labels that guide care today and hopefully, in the future.
Our story begins in February 2012, when the crystal structure of human menin was stored in the Protein Data Bank under the unique ID 3U84. The path to this breakthrough was paved earlier by Jolanta Grembecka, PhD, Tomasz Cierpicki, PhD, and colleagues at the University of Michigan, who determined the structure of menin. While this was not human in origin, it was the first-ever menin protein structure to be solved and provided key insights that helped inform efforts to crystallize human menin.
Building on these foundational insights, the elucidation of the human menin structure represented a critical advance. Those efforts were funded in part by an American Association for Cancer Research (AACR) grant in partnership with the Caring for Carcinoid Foundation (now Neuroendocrine Tumor Research Foundation), which was awarded in 2011 to Xianxin Hua, MD, PhD, now a professor of cancer biology at the University of Pennsylvania. This discovery meant that scientists could see the three-dimensional shape of human menin for the first time—they saw regions in menin that could explain its biology when it is bound to other molecules.
Hua, who had been interested in menin since the gene for it—MEN1—was first cloned in 1997, explained that the rationale behind his research that fetched the grant was based on MEN1 genetics. “We knew that patients who inherit a defective copy of MEN1 develop tumors of the pancreas, pituitary, and parathyroid,” he explained. “We wanted to understand how menin worked in these tissues, with the hope that this knowledge would lead to new therapies.”

Basic Research Uncovers Menin’s Connection to Leukemia
Hua’s research explained how one of the grooves on menin’s surface, which was uncovered in collaboration with Ming Lei, PhD, then at the University of Michigan, could recognize and grab onto short stretches of amino acids on the proteins called MLL1 or transcription factor JunD. While the grant targeted neuroendocrine tumor research, the rationale of Hua’s work rested on menin’s role as a scaffold protein that regulates transcription and signaling across tissues—a role that would later define leukemia biology.
Hua and colleagues added to this structural story of menin at the AACR Annual Meeting 2012, by showing that, when complexed with other oncoproteins, it keeps immature blood cells stuck in an undeveloped state—preventing them from maturing into normal blood cells. Menin was now “visible” to the scientific community, and its unveiling showed how funding for one tumor type spilled into mechanistic understanding relevant to another.
Once the picture of menin existed in three dimensions, researchers in the drug development community went to work. In 2015, a team of researchers led by Grembecka and Cierpicki reported the first potent, orally bioavailable small‑molecule inhibitors of the menin-MLL interaction—molecules like MI‑503 and MI‑463 that bound menin, lowering HOXA/MEIS1 gene expression, and prolonging survival in mouse models of leukemia without harming normal blood cell production. This was evidence that menin was viable as a drug target and selective enough to leave normal stem cell processes largely intact.
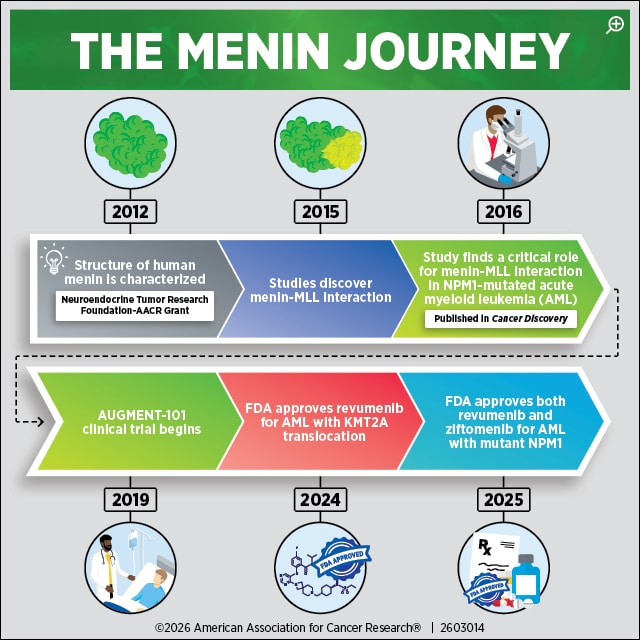
By 2016, structure‑based design studies had refined thienopyrimidine scaffolds—a class of small molecules—into compounds like MI‑538, with single‑digit‑nanomolar affinity for menin and strong cellular activity in MLL‑AF9 leukemia models, highlighted in an AACR Annual Meeting abstract. The study showed how connecting multiple individually optimal chemical moieties onto a single base molecule did not produce a better drug; in this case, it degraded the molecules’ pharmacokinetics and polarity. It meant researchers could not just cobble together a drug that would be the “magic bullet” to bind to menin perfectly, but it did further the understanding of how the protein worked.
Translating the Discovery to NPM1-mutant Acute Myeloid Leukemia
Up until this point, the translational rationale for menin inhibition centered on KMT2A‑rearranged leukemias. In late 2016, Scott A. Armstrong, MD, PhD, and collaborators provided rigorous evidence published in the AACR journal Cancer Discovery that NPM1‑mutant acute myeloid leukemia (AML)—the most common form of adult AML—also depends on the menin-MLL1 complex to maintain aberrant HOX/MEIS1 gene expression.
Using gene-editing experiments and pharmacologic tools, they showed that blocking menin-MLL1 releases the differentiation block, a state where blood cells are unable to mature, in NPM1‑mutant cancer cells. This was the first study that showed how menin could be targeted beyond the relatively rare KMT2A-rearranged leukemia patient population, taking it from a niche strategy into a potential mainstay for a large segment of adult AML. The AACR Annual Meeting 2016, for which Armstrong was program chair, highlighted several investigations on menin, such as how SON, a nuclear protein, would interact with menin to inhibit menin-MLL complex formation.
From there, clinical translation followed. In 2019, the AUGMENT‑101 phase I/II, multicenter trial enrolled adult and pediatric patients with relapsed or refractory acute leukemias, including KMT2A translocations and NPM1 mutations. Patients received revumenib, an oral inhibitor of the menin-KMT2A interaction, until disease progression, unacceptable toxicity, lack of morphological leukemia-free response after four cycles of treatment, or receipt of a stem cell transplant. Overall, 21.2% of patients experienced a complete remission or a complete remission with partial hematopoietic recovery (CR/CRh), meaning no signs of cancer remained, but abnormalities in blood cell counts persisted.
Revumenib Becomes the First FDA-approved Menin Inhibitor, and More Approvals Followed
Data from AUGMENT-101 led to the first regulatory win for targeting menin—in November 2024, the FDA approved revumenib under the brand name Revuforj for patients with relapsed or refractory acute leukemia with a KMT2A translocation. Less than a year later, in October 2025, the FDA expanded revumenib’s label to include relapsed or refractory AML with a susceptible NPM1 mutation in adult and pediatric patients 1 or older who have no satisfactory alternatives.
This progress in successfully translating the science behind targeting menin has had real, tangible results. Jack Moorman, diagnosed in 2023 with AML and a KMT2A translocation, entered a study combining revumenib with azacitidine and venetoclax (Venclexta) and achieved complete remission after one cycle, then underwent a stem cell transplant that reshaped his long‑term odds. His story appeared in the AACR Cancer Progress Report 2025 and stands as a reminder that investigating novel mechanisms is often the difference between holding disease at bay long enough to reach potentially curative therapy and watching relapse outrun induction.
Tyler Peryea, a teenager whose cancer journey is featured in the AACR Pediatric Cancer Progress Report 2025, was diagnosed with AML in 2024. Since the cancer had a rare mutation pattern, he could not be treated with standard options and was referred to Dana‑Farber/Boston Children’s Cancer and Blood Disorders Center for a menin inhibitor trial. The treatment eventually allowed Tyler to get a stem cell transplant, and he continues to take revumenib to prevent a relapse.
While revumenib had its time in the spotlight as the first menin inhibitor, ziftomenib (Komzifti) advanced in parallel as another drug in this class. Ziftomenib was evaluated in the KOMET‑001 clinical trial, with CR/CRh observed in 21.4% of evaluable patients in the trial.
The results from KOMET-001 led to the FDA approval of ziftomenib for adults with relapsed or refractory NPM1-mutant AML who had no satisfactory alternative treatment options, consolidating menin inhibition as a class with two approved therapeutic agents.
Expanding Horizons in Menin Inhibition
The march of scientific advancement involving menin continues. Two randomized, double‑blind studies in the KOMET-017 trial are testing ziftomenib in frontline combinations with venetoclax and azacitidine as well as different combinations of cytarabine and daunorubicin for patients with either NPM1‑mutant or KMT2A‑rearranged AML. This trial design is geared towards understanding whether adding menin inhibition improves event‑free survival and deepens minimal residual disease (MRD)‑negative remission.
Another study comes at menin from the immunotherapy angle. Investigators have shown that menin inhibition can upregulate CLEC12A (CLL‑1) on leukemia cells, potentially priming them for CLL‑1-directed chimeric antigen receptor (CAR) T-cell therapy in NPM1‑mutant and KMT2A‑rearranged AML. If confirmed clinically, menin inhibitors could become the first step in “prime‑and‑target” therapeutic sequences where a small molecule prepares the tumor for a cellular therapy that finishes the job.
The connection between chromatin adaptor disruption and antigen presentation on the leukemia cell surface suggests the field’s next frontier might be the wedding of targeted and immune therapies, not merely their co‑administration. Hua, who has observed these developments over nearly three decades, sees the field as still very much in its early chapters.
“The inhibitors by themselves have median response rates at about two to five months—while there is a lot to deal with in this approach, combining immunotherapy with menin inhibition to make the response duration extend much longer, from five months to two or three years, or get a remission deep enough to proceed to bone marrow transplantation would be a great direction to move forward,” he noted.
As of 2026, AACR journals alone index more than 600 publications on menin, underscoring how research on menin and its inhibitors has accelerated since the protein proved targetable. According to Hua, menin studies have been making headway in cancers other than leukemia as well. Analysis of patient data from The Cancer Genome Atlas shows that menin is expressed at higher levels in colorectal tumors than in normal tissue, and work from Hua’s laboratory has shown that blocking menin relieves its repression of insulin gene transcription and insulin-producing β cell proliferation in the pancreas—a strategy with therapeutic potential being tested for type 1 diabetes in the COVALENT-112 trial and type 2 diabetes in the COVALENT-111 trial.
All this progress started with a picture—a picture produced by scientists who were just trying to understand the structure of a protein called menin.
This was a story about how studying a protein-protein interaction led to FDA-approved treatments. This was a story about how funding fundamental research in its infancy ultimately produced life-changing innovations for patients. This was a story about the power of discovery science.
For more fundamental research that shows great potential, read the “From the Bench” series of posts released quarterly.