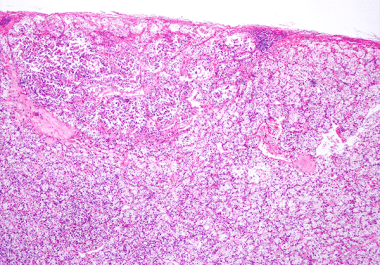
Full Approval for a Bladder Cancer Treatment
Enfortumab vedotin-ejfv plus pembrolizumab was approved for certain bladder cancers. The U.S. Food and Drug Administration (FDA) has approved enfortumab vedotin-ejfv (Padcev) plus pembrolizumab (Keytruda) for the treatment of patients with locally advanced or...