
Coordination of Care
Pilot program emphasizes standards to streamline patient care.


Pilot program emphasizes standards to streamline patient care.

Expansions in coverage and eligibility through the Affordable Care Act have made Medicaid a lifeline for many people with cancer, but benefits vary from state to state.
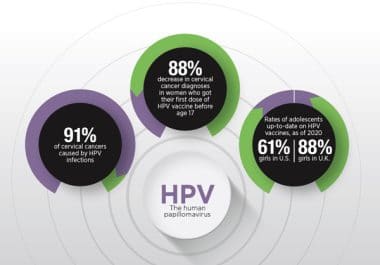
Sixteen years after the introduction of the HPV vaccine, research sees an effect on cervical cancer rates in young women.
FDA approves combination immunotherapy for treating melanoma, and screening tool improves pancreatic cancer detection.

Different types of colorectal polyps can be difficult to distinguish, a new AI program improved pathologists' accuracy in a recent study.

Research suggests short bursts of intense calorie restriction could improve treatment response in patients with a range of cancers.

Medical oncologist Lola Fashoyin-Aje describes the Food and Drug Administration’s efforts to make clinical trials more inclusive.

Success of immune checkpoint inhibitors in clinical trials does not always carry over to patients in the clinic.

Is it the beginning of the end or the end of the beginning?

Many cancer patients have problems with memory or thinking that can linger for years after treatment. The cause is a mystery, but new tactics are helping many people cope with its effects.