Combining Targeted Therapies for Lung Cancer
The FDA approved a combination of molecularly targeted therapeutics for use in combination to treat patients with metastatic non-small cell lung cancer.


The FDA approved a combination of molecularly targeted therapeutics for use in combination to treat patients with metastatic non-small cell lung cancer.

Women who work the night shift may have an increased risk of breast, skin, and gastrointestinal cancers, according to a study.

A recent study revealed large variations in colorectal cancer screening rates among states and counties. Colorectal cancer is the fourth most commonly diagnosed cancer in the United States with more than 135,000 diagnoses and...

The May 2018 FDA approval of the immunotherapeutic durvalumab makes it the first treatment approved to reduce the risk of stage 3 lung cancer progressing.
The FDA approved a hormone therapy for treating men with nonmetastatic prostate cancer that has stopped responding to standard hormone therapy.
The FDA expanded the use of a molecularly targeted therapeutic to include the prevention of bone complications in patients with multiple myeloma.
The FDA approved a molecularly targeted therapeutic for treating certain patients who have breast cancer that tests positive for a cancer-associated BRCA1 or BRCA2 mutation.

An American Association for Cancer Research (AACR) Special Conference highlighted the connections between weight and cancer.
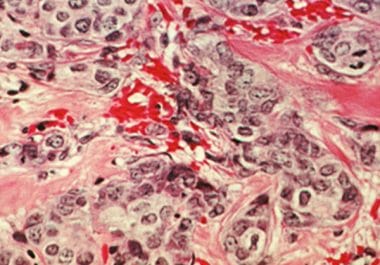
Whole-genome sequencing may help identify the patients who are most likely to benefit from certain types of chemotherapy.

Thyroid cancer survivors – especially those diagnosed before age 40 – have elevated risk for several aging-related diseases.