
New Treatment Approved for Lung Cancer
KRAS mutations were once thought to be resistant to drug therapy.


KRAS mutations were once thought to be resistant to drug therapy.
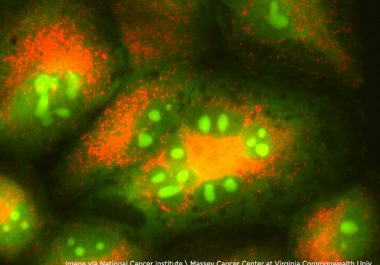
The FDA granted accelerated approval to a new targeted therapeutic and companion diagnostic test for certain adult patients with lung cancer. The U.S. Food and Drug Administration (FDA) granted accelerated approval to mobocertinib (Exkivity)...
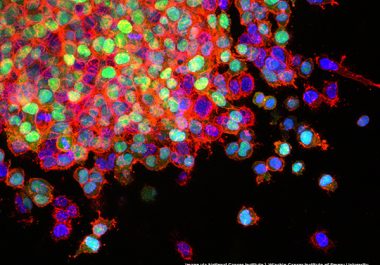
The U.S. FDA has granted accelerated approval to the first KRAS inhibitor, along with two companion diagnostics, for certain patients with KRAS-mutated lung cancer. The U.S. Food and Drug Administration (FDA) granted accelerated approval...
The FDA granted accelerated approval to the first targeted therapeutic and a companion diagnostic for certain adult patients with non-small cell lung cancer that tests positive for specific mutations in a protein that helps...
Lung cancer screening recommendations expand to include more smokers, and more news of the week from Cancer Today.
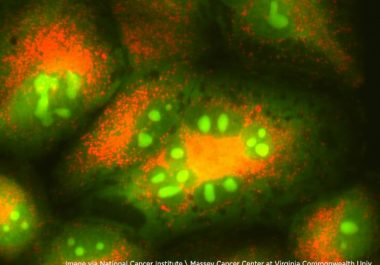
The FDA has approved an immune checkpoint inhibitor – a type of cancer immunotherapy – as a first-line therapeutic for certain patients with non-small cell lung cancer.
Tool outperformed preexisting mathematical models and a previously established artificial intelligence tool

The FDA granted accelerated approval to a new oral targeted therapeutic to treat certain adult patients with the most common form of lung cancer.

Researchers in Japan observe the rare occurrence of a mother passing cancer cells to their child, and more news of the week from Cancer Today.
Study finds adherence to follow-up screening guidelines is low among people at high risk of developing lung cancer, and more news of the week from Cancer Today.