August 2: The Week in Cancer News
Blood test approval widens access to colorectal cancer screening option, and language barriers interrupt cancer care before it starts.


Blood test approval widens access to colorectal cancer screening option, and language barriers interrupt cancer care before it starts.
Afamitresgene autoleucel has received accelerated approval to treat certain patients with synovial sarcoma. The U.S. Food and Drug Administration (FDA) has granted accelerated approval to afamitresgene autoleucel (Tecelra, afami-cel) for the treatment of certain...
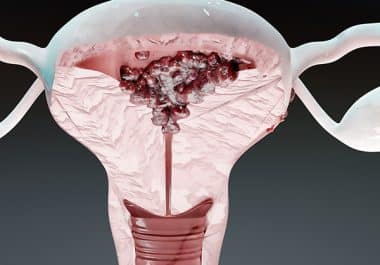
The FDA has approved dostarlimab-gxly plus chemotherapy to treat more patients with endometrial cancer. The U.S. Food and Drug Administration (FDA) has approved dostarlimab-gxly (Jemperli) in combination with the chemotherapies carboplatin and paclitaxel, followed...
The targeted therapy was approved by the FDA for use in combination with bortezomib, lenalidomide, and dexamethasone. The U.S. Food and Drug Administration (FDA) has approved daratumumab and hyaluronidase-fihj (Darzalex Faspro), plus bortezomib, lenalidomide,...
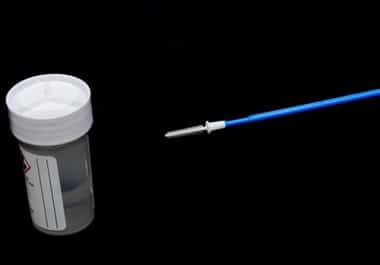
New cervical cancer self-screening options and a portable blood test device could lead to changes in at-home cancer screening and monitoring.
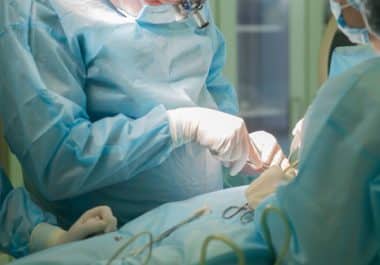
Double mastectomy doesn’t lead to a survival advantage over lumpectomy or single mastectomy, and research explores hearing loss linked to cisplatin.

Clinical trials for dogs with cancer can extend the lives of our four-legged friends while revealing insights about cancer in humans.

The AACR journal editors' picks for July 2024 include a review of the risks from CAR T-cell therapy, advances made in sarcoma, and more.

Learn how liquid biopsy could be used to detect cancer and guide treatment as researchers work to develop more noninvasive screening options.
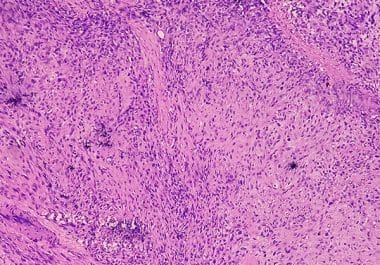
Researchers are making progress on genetic classification of sarcoma tumors, the use of T-cell therapy, and nanomedicine delivery.