Use of Nivolumab Expanded to Sixth Cancer Type
Last week, the U.S. Food and Drug Administration (FDA) announced that it had approved expanding the use of the immunotherapeutic nivolumab (Opdivo) to include the treatment of certain patients with bladder cancer. Specifically, the FDA approved nivolumab for treating patients with locally advanced or metastatic urothelial carcinoma whose disease has progressed despite treatment with platinum-based chemotherapy or whose disease progressed within 12 months of neoadjuvant or adjuvant treatment with platinum-based chemotherapy.
With this decision, nivolumab is now approved for treating six types of cancer.
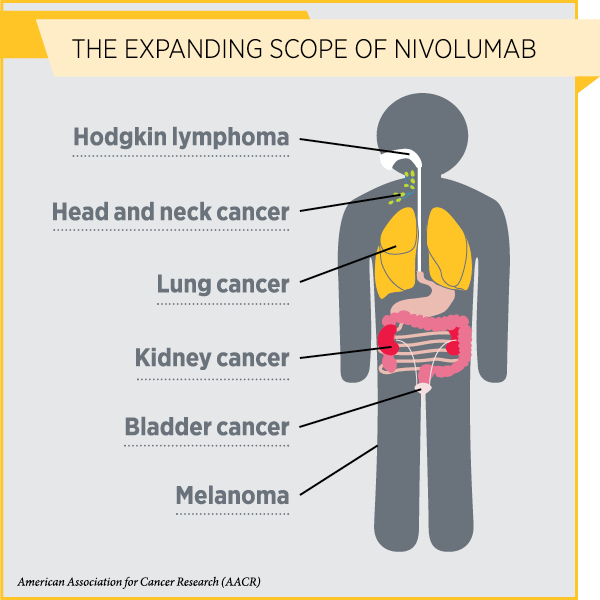
Bladder cancer is expected to be the sixth most commonly diagnosed cancer in the United States in 2017, with an estimated 79,030 new cases of the disease. With 16,870 people anticipated to die from the disease in 2017, it will be the ninth most common cause of cancer-related death in the United States.
More than 90 percent of bladder cancers diagnosed in the United States begin in urothelial cells that line the inside of the bladder; they are referred to as urothelial carcinomas.
The approval of nivolumab for urothelial carcinoma was based on results from the phase II CheckMate-275 clinical trial, according to Bristol-Meyers Squibb (BMS), the company that manufactures nivolumab. In brief, the results showed that 19 percent of the 270 patients who received nivolumab had an objective response for a median of 10.3 months. Of the 53 patients who responded, seven had a complete response and 46 had a partial response.
Given that the approval of nivolumab centers on response data, rather than overall survival, BMS is required by the FDA to conduct additional studies to confirm that the immunotherapeutic improves survival for patients with urothelial carcinoma.
Nivolumab works by releasing a brake (or checkpoint protein) called PD-1 on cancer-fighting immune cells called T cells. Nivolumab releases the PD-1 brake by preventing it from being engaged by two proteins called PD-L1 and PD-L2. Once the PD-1 brake is released by nivolumab, the T cells are able to destroy cancer cells.
Nivolumab is the second immunotherapeutic that targets the PD-1/PD-L1 braking system to be approved by the FDA for treating certain patients with urothelial carcinoma. As discussed on this blog, atezolizumab (Tecentriq), which targets PD-L1, was approved for this indication in May 2016, after it was shown to yield remarkable and durable responses for some patients, like Dave Maddison, who was featured in the AACR Cancer Progress Report 2016.
https://www.youtube.com/watch?v=U7dLmHP8zsk