Editors’ Picks, April 2026: Immunotherapy-boosting Bacteria, GLP-1 Agonists for Breast Cancer Survivors, and More
May flowers might be just around the corner, but April has at least one more shower in store: it’s raining new research, courtesy of this month’s Editors’ Picks. The fecund data from the April 2026 edition include a bacterium that may enhance immunotherapy for kidney cancer; targeted in vivo T-cell engineering for treating pancreatic cancer; overall survival implications for breast cancer survivors taking GLP-1 agonist drugs; and more. Clinical trial results have poured in as well—one clinical trial article for pediatric and adolescent and young adult tumors, and another for head and neck cancer.
Read the abstracts of the studies highlighted by the editors of the 10 peer-reviewed journals of the American Association for Cancer Research (AACR) below, where links to the full-text articles can also be found. Full-text access to these papers is freely available for a limited time.
Journal: Blood Cancer Discovery
Multiple myeloma is a malignancy of clonally expanded plasma cells shaped by complex interactions with the immune microenvironment (IME). To investigate immune correlates of treatment response and disease progression, we conducted multi-omics profiling including CD138neg single-cell RNA sequencing of 243 bone marrow samples from 102 patients (631,226 cells) and CD138pos bulk RNA and whole-genome sequencing from 209 samples. In longitudinal analyses, interferon-γ signaling associated with markers of impaired T-cell memory after autologous stem cell transplant, whereas naïve B-cell abundance and immunoglobulin diversity correlated with improved progression-free survival (HR = 0.48; P = 2.3e−4). At disease progression, multiple myeloma cells upregulated cancer–testis antigens (CTAg) and immune effector genes, with concurrent B-cell depletion, enrichment of myeloid-derived suppressor cell expression, and phenotypic T-cell exhaustion. These findings highlight dynamic immune–tumor interactions, identifying naïve B-cell reconstitution as a biomarker of durable response and CTAgs as potential targets for high-risk disease at progression.
Significance: Longitudinal profiling of multiple myeloma and the IME revealed dynamic immune–tumor interactions across the disease course. Altered expression in CD8+ T cells limited memory phenotype after transplant, whereas naïve B-cell recovery associated with sustained treatment response. At progression, CTAg expression associated with immunosuppression, revealing novel mechanisms of immune dysregulation.
This article was highlighted in the March issue.
Journal: Cancer Discovery

Somatic mosaicism is pervasively observed in human aging, with clonal expansions of cells harboring mutations in recurrently mutated driver genes. Bulk sequencing of tissues captures mutation frequencies but cannot reconstruct clonal architectures nor delineate how driver mutations affect cellular phenotypes. We developed single-cell genotype-to-phenotype sequencing (scG2P) for high-throughput, highly multiplexed, joint capture of genotyping of mutation hotspots and mRNA markers. We applied scG2P to aged esophagus samples from six individuals and observed large numbers of clones with a single driver event, accompanied by rare clones with two driver mutations. NOTCH1 mutants dominate the clonal landscape and are linked to stunted epithelial differentiation, whereas TP53 mutants promote clonal expansion through both differentiation biases and increased cell cycling. Thus, joint single-cell highly multiplexed capture of somatic mutations and mRNA transcripts enables high-resolution reconstruction of clonal architecture and associated phenotypes in solid tissue somatic mosaicism.
Significance: Joint single-cell capture of somatic mutations and mRNA transcripts reconstructs clonal architecture and associated phenotypes of the phenotypically normal esophagus, providing the first single-cell genotype–phenotype map of this clonally mosaic tissue to accelerate our understanding of human somatic evolution in solid tissues and provide a window into early cancerous states.
This article was featured on the cover of the April issue, where it was highlighted.
Journal: Cancer Epidemiology, Biomarkers & Prevention
Background: Cervical cancer remains a significant health burden in China. National policies now allow human papillomavirus (HPV) DNA as a primary screening test, but many health resource–limited counties have not adopted it because programs cannot fund reagents or laboratory platforms. We conducted a multicenter study in 10 pilot counties where HPV DNA screening was introduced with project-supported reagents, platforms, and training.
Methods: We analyzed data from 10 pilot counties designated by the National Cervical Cancer Prevention Program. A total of 63,223 women aged 35 to 64 years were screened with three strategies: (i) cytology alone, (ii) HPV DNA testing with cytology triage, and (iii) HPV DNA testing with visual inspection with acetic acid/Lugol iodine (VIA/VILI) triage. Key indicators included screen positivity rate, colposcopy rate, cervical intraepithelial neoplasia (CIN) 2+ detection, positive predictive value (PPV), and number needed to refer (NNR). Inverse probability weighting was used to adjust for loss to follow-up.
Results: HPV-based strategies were superior to cytology across all indicators. CIN2+ detection was two to three times higher, with the highest PPV in the HPV + cytology group (21.9%) and the lowest NNR (5.73), indicating higher referral efficiency. Loss to follow-up rate was also significantly reduced under the HPV + VIA/VILI strategy. These advantages were most prominent among women aged ≥45 years.
Conclusions: Within the first year of implementation, HPV DNA–based screening is feasible and more effective in health resource–limited areas of China and flexible triage models can be adapted.
Impact: The findings support the integration of HPV DNA testing into national cervical cancer screening programs and highlight needs for robust follow-up systems in underserved populations.
This article was featured on the cover of the April issue, where it was also highlighted.
Journal: Cancer Immunology Research
Fibroblast activation protein (FAP), which is highly expressed on cancer-associated fibroblasts (CAF), is a promising therapeutic target to achieve normalization of the tumor microenvironment. We previously established an ex vivo retroviral-transduced FAP-specific chimeric antigen receptor (FAP-CAR) T-cell approach to deplete FAP+ CAFs that resulted in delayed tumor growth associated with disruption of desmoplastic matrix and enhanced immune cell infiltration and reversed immune exclusion and immunosuppression. In this study, we describe an in vivo strategy for generating FAP-CAR T cells using anti-CD5–conjugated targeted lipid nanoparticles (tLNP) encapsulating FAP-CAR mRNA and assessed the efficacy of this approach compared with adoptive transfer of retrovirus-transduced CAR T cells in a preclinical model of pancreatic ductal adenocarcinoma. With transient CAR expression in >45% of splenic, >69% of circulating, and >35% of tumor-infiltrating T cells, the abundance of peripheral and intratumoral FAP-CAR+ T cells detected following a single intravenous dose of FAP-CAR mRNA tLNPs was greater than that detected following administration of 1 × 107 ex vivo retrovirally transduced FAP-CAR T cells. Furthermore, in vivo mRNA CAR T-cell engineering resulted in as good or greater inhibition of tumor growth as compared with adoptive transfer of ex vivo retroviral-engineered T cells. Given that in vivo generation of CAR T cells resulted in transient CAR expression and circumvented the need for autologous T-cell isolation, viral vectors, and lymphodepletion, this platform represents a potentially safer, more accessible, and cost-effective method for targeting stromal cells to normalize the tumor microenvironment in desmoplastic tumors and has potential implications for tumor antigen–targeted CAR T cells.
Journal: Cancer Prevention Research
Integrative Analysis of Multiomic Pathways Predicts Cancer-Affected Lobes in Lung Cancer
Lung cancer is the leading cause of cancer-related deaths. The human microbiome plays an important role in regulating response to cancer therapeutics, outcomes, and biological processes. However, little is known about the interplay between the lung microbiome and other biological processes in cancer. In an exploratory pilot study, we collected bronchoalveolar lavage fluid and brushings from 20 patients with early-stage lung cancer and performed microbial sequencing, untargeted metabolomics, and cytokine analysis. In addition, we employed computational and machine learning approaches to identify integrated microbial–immunometabolic pathways. Finally, we performed preliminary mechanistic studies to confirm our findings. Previously, we published that upper airway microbiota were selectively enriched in tumor-affected lobes. In the present study, we demonstrate that enrichment of protumorigenic cytokines and specific fatty acids is associated with tumor-affected lobes. Finally, we find that long-chain fatty acid stimulation of macrophages leads to neoplastic transformation of lung epithelial cells. Therefore, the findings of this study identify a perturbed fatty acid–macrophage axis that is a potential biomarker of early-stage lung cancer and will lead to the development of novel therapeutic agents.
Prevention Relevance: This study identifies a lung microbiome-driven immunometabolic axis involving stearic acid and MIP1β in tumor-affected lobes of patients with early-stage lung cancer. These localized microbial and cytokine–metabolite signatures may serve as biomarkers for early detection and provide targets for preventive strategies in high-risk individuals undergoing lung cancer screening.
Journal: Cancer Research (April 1 issue)
Transcriptomic Plasticity Is a Hallmark of Metastatic Pancreatic Cancer
Metastasis is the leading cause of cancer deaths. To develop strategies for intercepting metastatic progression, a better understanding of how tumor cells adapt to vastly different organ contexts is needed. To investigate this question, a single-cell transcriptomic atlas of primary tumors and diverse metastatic samples (liver, omentum, peritoneum, stomach wall, lymph node, and diaphragm) from a patient with pancreatic ductal adenocarcinoma who underwent rapid autopsy was generated. Using unsupervised archetype analysis, both shared and site-specific gene programs were identified, including lipid metabolism and gastrointestinal programs prevalent in peritoneal and stomach wall lesions, respectively. We developed phylogenetic inference from copy-number alterations in single-cell sequencing observations (PICASSO) as a probabilistic approach for inferring clonal phylogeny from single-cell and matched whole-exome sequencing data. Comparison of PICASSO-generated clonal structure with phenotypic signatures revealed that pancreatic cancer cells adapted to local environments with minimal contribution from clonal genotype. Our results suggest a paradigm whereby strong environmental effects are imposed on highly plastic cancer cells during metastatic dissemination.
Significance: Single-cell transcriptional profiling of primary tumor and metastases from rapid autopsy samples of an individual with pancreatic cancer, combined with probabilistic clonal inference by PICASSO, reveals substantial transcriptomic plasticity in metastatic cells.
This article is part of a special series: Driving Cancer Discoveries with Computational Research, Data Science, and Machine Learning/AI.
Journal: Cancer Research (April 15 issue)
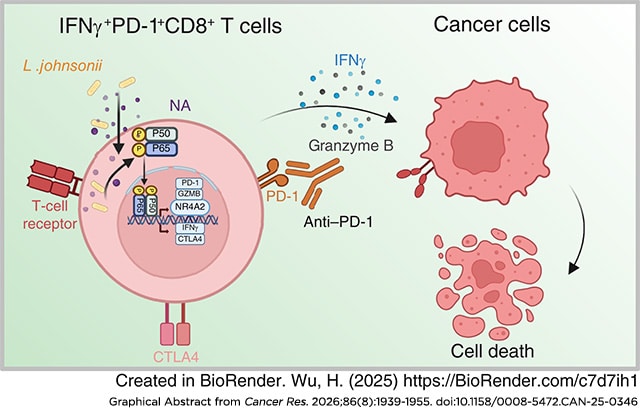
Although surgical resection is an effective intervention for early-stage hepatocellular carcinoma (HCC), postoperative recurrence remains a major clinical hurdle. Delving into the mechanisms underlying relapse and pinpointing potential therapeutic targets are imperative for improving outcomes for patients with HCC. By comparing the microbiota composition in patients with early- and non-relapsing HCC, we identified that Lactobacillus was enriched in patients with relapse-free HCC, serving as an independent prognostic predictor of disease-free survival. Higher levels of intratumoral Lactobacillus johnsonii correlated with an increased abundance of IFNγ+PD-1+CD8+ T cells. Single-cell RNA sequencing, transcriptomic profiling of intratumoral CD45+ immune cells, and in vitro functional assays demonstrated that L. johnsonii preferentially enhanced this cytotoxic-exhausted T-cell population. Nicotinic acid (NA) served as a key metabolite derived from L. johnsonii that expanded IFNγ+PD-1+CD8+ T cells and upregulated effector (granzyme B) and exhaustion (CTLA4) markers. Mechanistically, both L. johnsonii and NA activated the NF-κB pathway, leading to increased IFNγ production and upregulation of the transcription factor NR4A2, which in turn sustained PD-1 expression on CD8+ T cells. Combining L. johnsonii or NA with anti–PD-1 therapy synergistically inhibited tumor relapse and tumor growth in immunocompetent or humanized mice. Crucially, the antitumor efficacy of L. johnsonii was CD8+ T cell–dependent, as depletion abolished its activity. This work unveils a mechanism by which L. johnsonii and its metabolite NA enrich intratumoral IFNγ+PD-1+CD8+ T cells, thereby reshaping the immune microenvironment to potentiate immunotherapy efficacy and suppress HCC recurrence.
Significance: Lactobacillus johnsonii produces nicotinic acid that expands intratumoral IFNγ+PD-1+CD8+ T cells, which prevents hepatocellular carcinoma relapse and enhances the efficacy of anti-PD-1 therapy.
Journal: Clinical Cancer Research (April 1 issue)
Purpose: Arm D of the AcSé-ESMART proof-of-concept phase I/II platform trial aimed to define the recommended phase II dose (RP2D), pharmacokinetics, activity, and biomarkers of the PARP inhibitor olaparib with irinotecan in pediatric patients with recurrent/refractory malignancies.
Patients and Methods: Olaparib was administered orally twice daily on days 1 to 10 and irinotecan intravenously on days 4 to 8 of a 21-day cycle. Dose escalation followed the continuous reassessment method; activity was assessed in diverse tumor types (cohort 1) and Ewing sarcoma (cohort 2) according to a minimax Simon 2-stage design. Cohorts were enriched for alterations in homologous recombination repair (HRR) pathways.
Results: Seventy patients (median age, 14.9 years; range, 5.0–23.8) were included, 34 with diverse tumor types (25 with HRR gene alterations) and 36 with Ewing sarcoma. Sixty-six patients received 348 treatment cycles (median, 2; range, 1–51) over four dose levels. Main toxicities were gastrointestinal and myelosuppression; the RP2D was olaparib 90 mg/m2 twice daily and irinotecan 20 mg/m2/day. Olaparib exposure in children was equivalent to that in adults. The overall response rate was 9.1% (cohort 1, 11.8%; cohort 2, 6.3%). Four patients with osteosarcoma, pineoblastoma, choroid plexus carcinoma, and neuroblastoma experienced a partial response and were treated for nine to 51 cycles. Two patients with Ewing sarcoma experienced a complete and a partial response for 10 and 42 cycles, respectively. Genetic analyses suggest a high aneuploidy score possibly associated with objective response and prolonged stable disease.
Conclusions: Olaparib combined with irinotecan demonstrated activity in pediatric tumors, which was enriched among tumors that exhibited aneuploidy.
Journal: Clinical Cancer Research (April 15 issue)
Purpose: This multicenter, phase IIa trial (NCT04868162) investigated the efficacy and safety of becotatug vedotin, an anti–epidermal growth factor receptor (EGFR) antibody–drug conjugate, in patients with recurrent or metastatic squamous cell carcinoma of the head and neck (R/M SCCHN) who have limited therapeutic options.
Patients and Methods: Patients with R/M SCCHN who progressed after platinum-based chemotherapy and/or programmed cell death 1 (PD-1)/programmed death ligand 1 (PD-L1) inhibitors, including those with multiple lines of prior therapy, were administered intravenous becotatug vedotin at either 2 or 2.3 mg/kg every 3 weeks. The primary endpoint was the objective response rate (ORR).
Results: Sixty-seven patients were enrolled (35 received 2 mg/kg and 32 received 2.3 mg/kg). The ORR was 20.9% [14/67, 95% confidence interval (CI), 11.9–32.6], with a median duration of response (DoR) of 10.9 months (95% CI, 2.6–15.1). The median progression-free survival (PFS) was 2.9 months (95% CI, 1.8–3.9), and the median overall survival (OS) was 6.7 months (95% CI, 5–8.9). Treatment-related adverse events (TRAE) were reported in 91% (61/67) patients, most commonly being rash (26.9%), pruritus (25.4%), constipation (23.9%), and anemia (20.9%).
Conclusions: Becotatug vedotin demonstrated promising antitumor activity with a manageable safety profile in previously heavily treated R/M SCCHN, particularly at the recommended dose of 2.3 mg/kg, among patients who had failed platinum-based chemotherapy and PD-1/PD-L1 inhibitors (≤2 prior lines of therapy).
Journal: Molecular Cancer Research
Although kidney cancer arises from a variety of nephron cells with diverse characteristics and develops intratumor heterogeneity, a model to elucidate these complexities is incompletely developed. In this study, we report a genetically engineered human induced pluripotent stem cell–derived kidney organoid (HKO) model, which may recapitulate an early stage of renal tumorigenesis. When we overexpressed the VCL-ALK fusion gene, a renal oncogene, in HKO, tubular cells proliferated, and this proliferation was sustained under long-term culture or hypoxic conditions. In addition, the proliferating tubular cells migrated into the renal parenchyma of host mice upon transplantation. The deconvolution analysis and IHC revealed that the proliferating tubular cells could be arrested at an immature tubular progenitor stage with increased expressions of LIM homeobox protein 1 and jagged canonical Notch ligand 1, critical regulators of nephrogenesis. Our HKO model advances our understanding of renal tumorigenesis in the context of the trajectory of nephron development.
Implications: Dysregulated nephron developmental machinery leads to aberrant cell proliferation of immature renal tubules.
This article was highlighted in the April issue.
Journal: Molecular Cancer Therapeutics
TPP-45142—an Anti-HER2 T-cell Engager—Designed for Selective HER2-Low Cancer Immunotherapy
The standard of care for patients with HER2-positive cancers is well established, but a significant unmet need exists for patients with HER2-low tumors, who do not meet the eligibility criteria for trastuzumab, and for patients with HER2-positive tumors, who are refractory to trastuzumab treatment. Therefore, in this study, we developed a NANOBODY domain–based HER2-targeting, T-cell receptor αβ–based T-cell engager (TCE) molecule—TPP-45142; it recognizes a HER2 epitope distinct from that recognized by trastuzumab and pertuzumab and redirects T cells to kill HER2-low cancers such as breast, gastric, and gastroesophageal junction adenocarcinoma cancers. TPP-45142 mediated potent T cell–dependent cytotoxicity against HER2-low cancer cell lines in vitro and inhibited in vivo tumor growth of HER2-low breast cancer xenografts. TPP-45142 was highly selective toward tumor cells expressing low HER2 levels than toward normal cardiac cells and exhibited a favorable therapeutic index as per a cytokine release assay. Thus, TPP-45142, with an improved safety profile, is a promising next-generation TCE for treating challenging HER2-low cancers.
This study was highlighted and featured on the cover of the April issue.
Journal: Cancer Research Communications
Weight Loss Patterns and Clinical Outcomes of GLP1 Receptor Agonists in Breast Cancer Survivors
The use of glucagon-like peptide-1 receptor agonists (GLP1-RA) for weight loss is increasing; implications in breast cancer survivors remain unclear. This retrospective cohort study evaluated treatment patterns, weight loss, and outcomes in breast cancer survivors receiving GLP1-RA at an academic institution. We evaluated patients with nonmetastatic [ductal carcinoma in situ (DCIS), stage 1–3] breast cancer who received GLP1-RA (2005–2024). Linear regression models estimated associations between weight change and clinical factors. After excluding DCIS, propensity score matching (1:2) was used to match patients who received GLP1-RA with patients who did not, based on confounding covariates. Kaplan–Meier estimates and log-rank tests compared disease-free survival (DFS) and overall survival (OS) between GLP1-RA users versus nonusers in the subgroup with invasive disease. We identified 1,022 patients; 79% had type 2 diabetes mellitus. The median weight and body mass index at GLP1-RA initiation were 86.8 kg (47.2–175 kg) and 33.5 kg/m2 (18.9–61.8 kg/m2). In semaglutide or tirzepatide users (442, 43.2%), the median weight change at 3, 6, and 12 months after GLP1-RA initiation was −1.9% (−13.2% to 14.9%), −3.1% (−20.2% to 19%), and −2.6% (−27.8% to 11.5%), respectively. Endocrine therapy and metformin use were associated with weight gain and loss, respectively; invasive disease stage was linked to greater weight loss. GLP1-RA was not associated with DFS, but OS significantly differed between GLP1-RA users (n = 810) and nonusers (n = 1,620; hazard ratio, 0.37; 95% confidence interval, 0.27–0.53; P < 0.0001). In conclusion, in this real-world study in breast cancer survivors, GLP1-RA was associated with modest weight loss and improved all-cause survival. Clinical trials are warranted to study GLP1-RA in this population.
Significance: This is the largest study describing real-world patterns of GLP1 receptor agonists in breast cancer survivors. Clinical trials should evaluate these agents for weight management as an adjunct to lifestyle interventions and the potential role in cancer control in breast cancer survivors.