
February 21: The Week in Cancer News
Men with metastatic prostate cancer urged to ask questions about care, and early-phase study suggests benefit of pancreatic cancer vaccine.


Men with metastatic prostate cancer urged to ask questions about care, and early-phase study suggests benefit of pancreatic cancer vaccine.

This month, the editors of AACR's journals show their love for studies about CAR T-cell efficacy, breast cancer risk, and more.

Radiation plus immunotherapy may help some patients with invasive bladder cancer keep their bladders. Patients with localized muscle-invasive bladder cancer (MIBC), or bladder cancer that has spread through the lining of the bladder and...
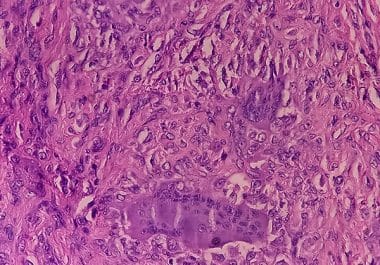
The FDA has approved a CSF1 small molecule inhibitor for certain patients with symptomatic tenosynovial giant cell tumor. The U.S. Food and Drug Administration (FDA) has approved vimseltinib (Romvimza) for the treatment of adult...

Pesticide firms try to block cancer-related lawsuits, and cancer pain leads to greater tobacco and cannabis use.
Researchers are examining ways to make treatments more targeted to each type of biliary tract cancer or to each patient.
The FDA has approved the antibody-drug conjugate brentuximab vedotin, plus lenalidomide and rituximab, for certain cases of pretreated large B-cell lymphoma. The U.S. Food and Drug Administration (FDA) has approved brentuximab vedotin (Adcetris), in...
The FDA has approved a new MEK inhibitor for certain patients with symptomatic neurofibromatosis type 1. The U.S. Food and Drug Administration (FDA) has approved mirdametinib (Gomekli) for the treatment of patients with neurofibromatosis...

Researchers explore musical toxicity from cancer treatment, and kidney cancer vaccine shows potential in early trial.

FDA approves an antibody-drug conjugate for people with HER2-ultralow breast cancer, and historic redlining practices have a lingering impact on cancer outcomes.