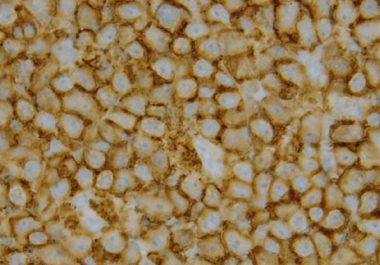
A New Molecularly Targeted Therapeutic for Non-Hodgkin Lymphoma
The FDA approved a molecularly targeted therapeutic for adults with mantle cell lymphoma that has progressed despite at least one prior treatment.


The FDA approved a molecularly targeted therapeutic for adults with mantle cell lymphoma that has progressed despite at least one prior treatment.
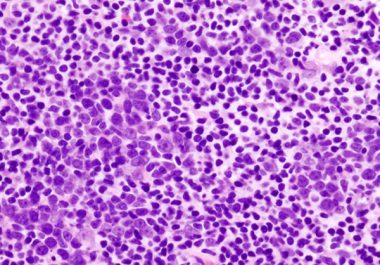
The FDA approved the second of a revolutionary new type of immunotherapy known as chimeric antigen receptor (CAR) T-cell therapy.
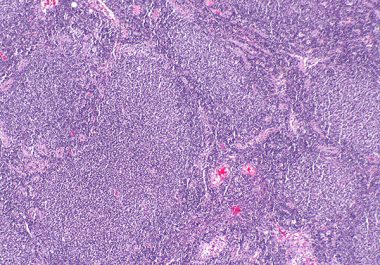
The FDA approved a molecularly targeted therapy for treating certain adults with follicular lymphoma, a form of non-Hodgkin lymphoma.
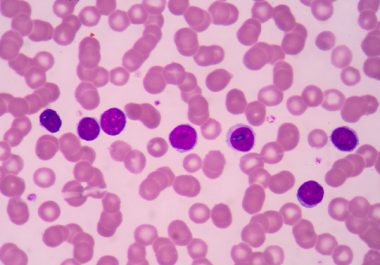
The FDA expanded the use of a targeted therapy for a form of non-Hodgkin lymphoma called marginal zone lymphoma.
The FDA expanded the use of an immunotherapy to treat certain patients with follicular lymphoma.

The FDA has approved of a treatment for Waldenström's macroglobulinemia, a rare form of non-Hodgkin lymphoma. Learn more.

A study found why some patients with mantle cell lymphoma develop resistance to the drug ibrutinib. The finding that could help researchers identify new treatment options. Mantle cell lymphoma is a rare type of...