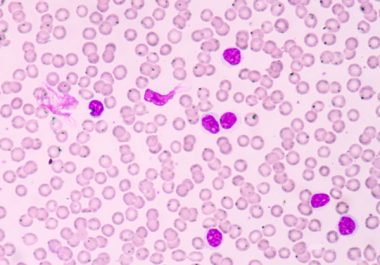
Targeted Therapy for Blood Cancer Patients
The FDA approved a molecularly targeted therapy to treat patients with chronic lymphocytic leukemia (CLL) and small lymphocytic lymphoma (SLL).


The FDA approved a molecularly targeted therapy to treat patients with chronic lymphocytic leukemia (CLL) and small lymphocytic lymphoma (SLL).
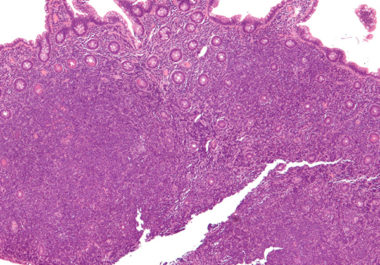
The FDA approved zanubrutinib to treat patients with mantle cell lymphoma, an aggressive type of non-Hodgkin lymphoma. The U.S. Food and Drug Administration (FDA) recently approved the molecularly targeted therapeutic zanubrutinib (Brukinsa) for the...
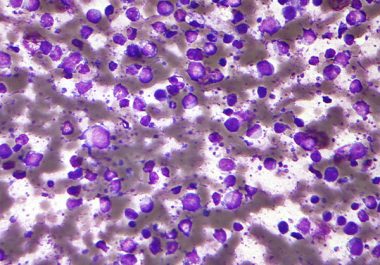
A molecularly targeted therapeutic has been approved to treat some patients with diffuse large B-cell lymphoma the most common type of non-Hodgkin lymphoma.
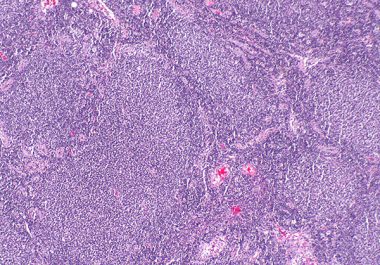
The FDA approved a new molecularly targeted therapeutic called for treating certain patients with blood cancer.
The FDA approved a combination of a molecularly targeted therapeutic and an immunotherapy for treating patients with Waldenström's macroglobulinemia.
The FDA approved a new immunotherapeutic for treating certain patients with two rare types of non-Hodgkin lymphoma.
The FDA expanded the use of the immunotherapy pembrolizumab for certain patients with an aggressive type of non-Hodgkin lymphoma.
The FDA expanded the use of the CAR T-cell therapy tisagenlecleucel to include certain patients with non-Hodgkin lymphoma.
The FDA expanded the use of a targeted therapy to include certain patients with two types of non-Hodgkin lymphoma.
The FDA approved a molecularly targeted therapeutic for adults with mantle cell lymphoma that has progressed despite at least one prior treatment.