Expanding Immunotherapy Options for Liver Cancer Patients
The FDA expanded the use of the immunotherapy pembrolizumab to include the treatment of certain patients with liver cancer.


The FDA expanded the use of the immunotherapy pembrolizumab to include the treatment of certain patients with liver cancer.
The FDA approved a molecularly targeted therapeutic for treating certain patients with metastatic non-small cell lung cancer.
The FDA approved a targeted therapeutic for treating patients with breast cancer that tests positive for a cancer-associated BRCA1 or BRCA2 mutations.
The FDA approved a molecularly targeted therapeutic for treating certain patients with non-small cell lung cancer.
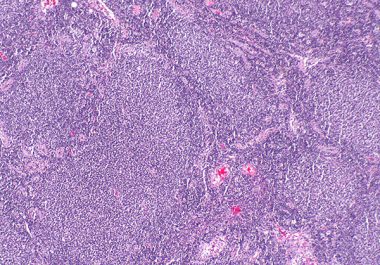
The FDA approved a new molecularly targeted therapeutic called for treating certain patients with blood cancer.
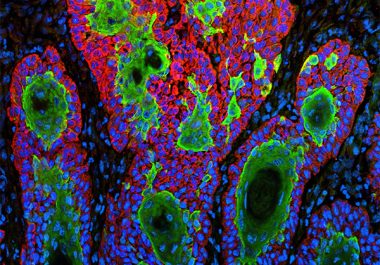
The FDA approved a new immunotherapeutic for treating patients with a type of skin cancer called cutaneous squamous cell carcinoma.

The FDA expanded the use of nivolumab to include treating certain patients with small cell lung cancer.
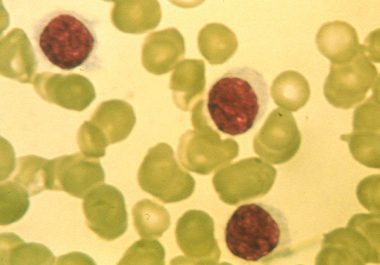
The FDA approved a new molecularly targeted therapeutic for treating certain patients with a rare form of leukemia called hairy cell leukemia.
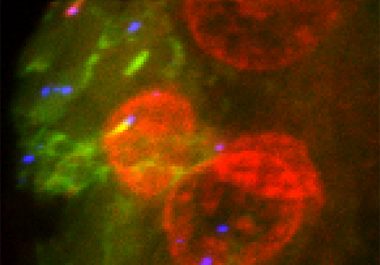
The FDA expanded the use of a molecularly targeted therapeutic to treat patients with the most common type of liver cancer, hepatocellular carcinoma.
The FDA approved a combination of a molecularly targeted therapeutic and an immunotherapy for treating patients with Waldenström's macroglobulinemia.