A New Type of Immunotherapy for Melanoma
A first-of-its kind immunotherapeutic has been approved by the FDA for the treatment of melanoma lesions in the skin and lymph nodes.


A first-of-its kind immunotherapeutic has been approved by the FDA for the treatment of melanoma lesions in the skin and lymph nodes.
The FDA has approved trabectedin for the treatment of certain patients with two types of soft tissue sarcoma, liposarcoma and leiomyosarcoma.
A nanotherapeutic form of the conventional chemotherapeutic has been approved by the FDA for treating certain patients with pancreatic cancer.
The FDA has expanded the use of an immune checkpoint inhibitor for the treatment of patients with lung cancer.

The FDA has approved a targeted therapy for certain patients with lung cancer.
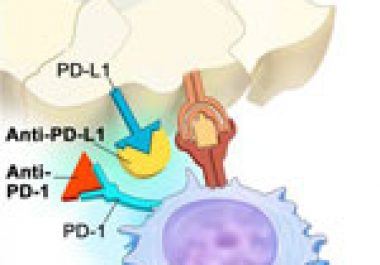
The FDA has expanded the use of pembrolizumab to include patients with advanced lung cancer that has progressed and whose tumors test positive for the protein PD-L1.
A new combination of cancer immunotherapeutics has been approved by the FDA for treating certain patients with melanoma.
A new combination of drugs was approved by the U.S. FDA for treating certain patients with advanced colorectal cancer whose disease has worsened despite treatment with chemotherapy and a biological therapy.

The FDA's approval of a targeted therapeutic called lenvatinib provides another option for patients certain forms of thyroid cancer.

The FDA has approved of a treatment for Waldenström's macroglobulinemia, a rare form of non-Hodgkin lymphoma. Learn more.