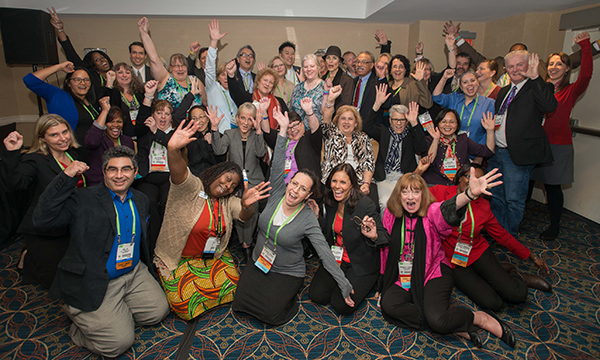Patients Power AACR Advocacy Efforts
Kim Hall Jackson is coming into her season.
Hall Jackson beat colorectal cancer in 2009, and has gone on to become deeply involved in the American Association for Cancer Research’s (AACR’s) Scientist↔ Survivor Program.
Her advocacy begins with her family. Colorectal cancer disproportionately affects African-Americans, and she wants her loved ones to know that screening is absolutely crucial.
“When we get together for Thanksgiving, Christmas, and Kwanzaa, I am the pain in the butt … literally,” she said, drawing laughs from her audience at a recent AACR advocacy meeting. “I will do whatever I need to do to make sure my loved ones don’t die because they were afraid of a test.”
On Nov. 16, 2015, the AACR hosted several dozen patient advocates and executives from leading pharmaceutical companies, as well as several cancer survivors who have participated in the Scientist↔Survivor Program. Their advocacy may begin with family and friends, but it extends well beyond, thanks in part to the scientific knowledge they’re building through the Scientist↔Survivor Program.
The program, founded in 1999, is designed to broaden survivor and patient advocates’ understanding of fundamental topics in cancer research and expose them to the cutting-edge science that is driving progress in the fight against cancer. A key component of the program is increasing the patient advocates’ scientific knowledge and supporting them as they share that new information with their home communities.
The Scientist↔Survivor Program provides participants with mentors like Emil Lou, MD, PhD, a gastrointestinal oncologist and an assistant professor in the department of hematology, oncology, and transplantation at the University of Minnesota.
“There is a much greater awareness in recent years that scientists need to come down from the ivory tower and interact meaningfully with patient advocates,” Lou told the audience.
Those connections happen every year, when participants in the Scientist↔Survivor Program attend the AACR’s Annual Meeting and the AACR Conference on the Science of Cancer Health Disparities in Racial/Ethnic Minorities and the Medically Underserved. There, they attend talks on cancer research that are tailored to a lay audience; they hold small-group meetings and roundtable conversations with scientific mentors; and they bring the concerns of the patient, survivor, and advocacy communities directly to scientists.

AACR Scientist↔Survivor Program Participant Susan Axler (left) with AACR Scientist↔Survivor Program Chair Anna Barker (right) at the 2013 AACR Annual Meeting. Photo by © AACR/Phil McCarten 2013.
Susan Axler, another participant in the program, told guests at the advocacy meeting that her experience with metastatic breast cancer has given her a strong desire to see patients’ needs reflected in every stage of cancer care, from the doctor’s office to the clinical trials where new developments are made. She cited an ambitious but intriguing idea for clinical trial design: “Ask 100 patients.”
Axler told guests of a clinical trial in California in which visiting nurses come to patients’ homes to draw blood, relieving them of the burden of travel when they’re not feeling well. As a patient advocate, she hopes to see a similar focus on the patient’s comfort and welfare in many more trials.
The AACR’s leadership treasures the time and experience of its patient advocates, and looks forward to many fruitful partnerships, according to Margaret Foti, PhD, MD (hc), chief executive officer of the AACR. “There is nothing that gives me greater joy than to work with our survivors and patient advocates,” Dr. Foti said.