Experts Forecast Cancer Prevention and Therapies in 2015
Since the beginning of the “war on cancer” in the 1970s, we have made consistent progress against cancer aided by paradigm-shifting technological advances. Last year, we witnessed significant developments being made on several different fronts. Proof of this is the approval by the U.S. Food and Drug Administration (FDA) of eight cancer drugs, including a few milestone “first-of-their-kind” drugs encompassing immunotherapies and targeted therapies, and a nine-valent vaccine that can prevent infections that cause cervical cancer.
The substantial gains we have made in cancer treatments are not without setbacks: For patients who respond to immunotherapies, it is the best treatment option available; however, immunotherapies are effective only in a limited population of cancer patients, and oncologists are still learning about the scope of cancers that immunotherapy can treat. When it comes to targeted therapies, the biggest problem to solve is drug resistance. However effective targeted therapies may be, most patients develop resistance to these drugs, and with many drugs this often happens within a year of treatment.
So the big question is, where do we go from here? What breakthroughs can we expect in 2015 that would impact cancer prevention and make modern medicine work better and for more patients?
We asked experts on three hot topics—immunotherapy, cancer genomics, and cancer prevention—to share their thoughts on what will be big this year. It is not unreasonable to expect a few more big leaps in 2015, experts say.
Immunotherapy in 2015

Drew Pardoll, MD, PhD, addresses participants in the Scientist↔Survivor Program at the AACR Annual Meeting 2014.
“Cancer immunotherapy is in the midst of an incredible growth spurt. I think there’s no reason to imagine that that’s not going to continue in 2015,” says Drew Pardoll, MD, PhD, professor of oncology, medicine, and pathology and co-director of the Cancer Immunology and Hematopoiesis Program at Johns Hopkins University. “This year will be the year in which anti-PD-1/PD-L1 immunotherapies—the immune checkpoint inhibitors—will be approved for a broad number of cancer types within one year of its first approval for melanoma,” he predicts.
The PD-1 checkpoint inhibitor, pembrolizumab, was approved by the FDA for the treatment of advanced melanoma in September 2014, which was followed by the approval of a similar drug, nivolumab, in December last year. Pardoll, who is also a senior editor of the AACR journal Cancer Immunology Research, foresees the approval of immune checkpoint inhibitors for lung, bladder, kidney, and head and neck cancers, as well as Hodgkin lymphoma, in 2015.
The next big thing to expect in immunotherapy this year is a lot of clinical trial activity and results with therapies using genetically modified T-cells, so-called chimeric antigen receptor T cell (CAR-T cell) therapies, says Pardoll. Cancer vaccines are not far behind, however. We can expect to see encouraging results from studies using cancer vaccines, such as the attenuated Listeria monocytogenes vaccine engineered to express human mesothelin, in pancreatic cancer and mesothelioma, Pardoll adds.
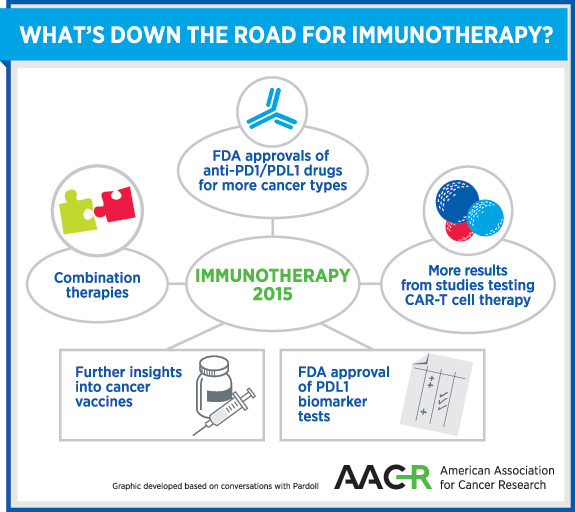
Given that the overall response rates with immunotherapy based on immune checkpoint inhibitors is around 20 percent, Pardoll says, “we still have a long way to go.” However, he also reminds us that the overall impact of immunotherapy as measured in years of cancer remission is about 30 times greater than that of all of oncogene mutation-targeted therapies combined.
The key to improving outcomes with immunotherapy lies in identifying the right patients for the right treatment. We can expect to see significant development on PD-L1 biomarker tests that can identify patients most likely to benefit from anti-PD-1/PD-L1 drugs, and lots of discussions around how to use these tests, says Pardoll. Another concept that we may hear more of in 2015, according to Pardoll, is adaptive resistance. PD-L1 expression by tumor cells suggests that a patient has an extant antitumor immune response. If a patient’s immune system does not have a strong enough antitumor response to induce PD-L1, a therapeutic maneuver to induce an antitumor immune response or amplify a weak one, such as a vaccine, will upregulate PD-L1. “So, one of the hot areas you’re going to see in 2015 is combinations of tumor vaccines with anti PD-1/PD-L1 drugs,” says Pardoll. “Combinations of checkpoint inhibitors will also be big in 2015,” he adds.
“I think that 2015 is going to be the year when you begin to see some of the first information about combinations of checkpoint inhibitors with targeted therapies, besides cancer vaccines. With combination therapies, we will have the opportunity to capture a larger number of patients into more durable response rates,” says Pardoll.
“With increasing momentum and interest from the investment community, 2015 is highly likely to be another exciting year for cancer immunotherapy,” Pardoll predicts.
Cancer Genomics in 2015
“With the power of cancer genomics, we have made a conceptual leap to the idea of tailoring therapy,” says William C. Hahn, MD, PhD, associate professor in the department of medicine at Harvard Medical School and director of the Center for Cancer Genome Discovery at Dana-Farber Cancer Institute. Cancer genomics has revolutionized the way we think about and treat cancer, and recent advancements and efforts are giving us the much-needed hope that we can use big data to better predict the disease.

William C. Hahn, MD, PhD, speaks at the 2013 AACR-NCI-EORTC International Conference on Molecular Targets and Cancer Therapeutics in Boston.
“There are three big directions for cancer genomics in 2015,” says Hahn, a senior editor of the AACR journal Molecular Cancer Research. “First, the question of how cancer genomes evolve with disease progression and treatment has not been deeply addressed. The large body of work that has been completed was focused on primary tumors—an essential starting point—but how cancers change over time is important to understand cancer better.
“The second direction is understanding cancer heterogeneity,” adds Hahn, who is also co-chair of the upcoming AACR Special Conference on Translation of the Cancer Genome. So far, we have learned that cancers are heterogeneous, and early work suggests that this heterogeneity is greater than initially imagined. Single cell-sequencing technologies will allow this to be explored in greater detail, he explains. Understanding cancer genomes will facilitate matching patients to therapies that they are likely to respond to, and will allow us to identify and anticipate resistance mechanisms. This knowledge also may enable us to predict side effects.
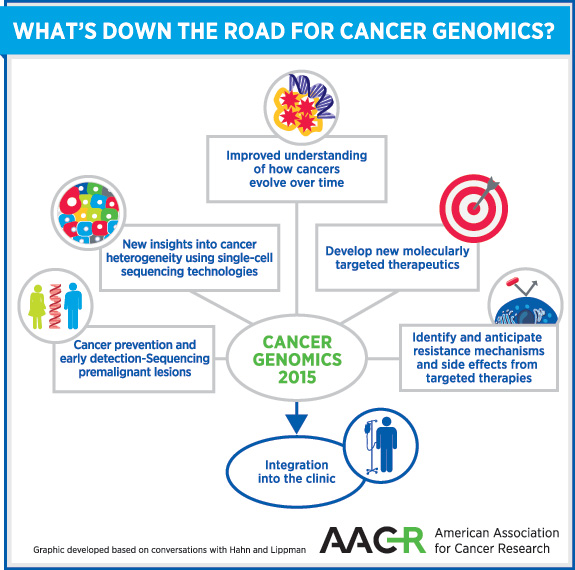
“We don’t know enough about cancer to reduce this to an engineering problem yet. Studying cancer genomes in patients will allow us to find ways to identify at-risk patients, and make more accurate and earlier diagnoses,” adds Hahn, emphasizing the utility of genomics in advancing cancer prevention and early detection.
“Finally, the ultimate direction for cancer genomics is integration into the clinic,” Hahn adds. He says this process has started but we are at the beginning, and likely to make significant progress this year.
Cancer Prevention in 2015

Scott Lippman, MD, is director of UC San Diego Moores Cancer Center and editor-in-chief of Cancer Prevention Research.
“The next big thing in cancer prevention is immunoprevention, specifically vaccines,” says Scott Lippman, MD, professor of medicine at UC San Diego, director of the UC San Diego Moores Cancer Center, and editor-in-chief of the AACR journal Cancer Prevention Research. He points us toward the impact of hepatitis B vaccines on liver cancer rates, and more recently, HPV vaccines on anogenital cancers and on oropharyngeal neoplasia. “Current and future efforts will include focus on discovering and targeting genetic drivers in premalignancy, including using tumor vaccines,” Lippman adds.
New methods of cancer screening, development of risk models that will identify high-risk individuals to improve risk-benefit ratio, and molecular imaging for early detection are other tremendously active areas of work, according to Lippman. The proposal by the Centers for Medicare and Medicaid Services in November last year to add annual low-dose CT screening for heavy smokers is an important step in early detection, Lippman notes.
“Moving toward personalized prevention using predictive biomarkers will be a major focus in 2015,” Lippman adds. Focused efforts on studying the biology of premalignancy using large-scale sequencing efforts similar to the TCGA study will be in the forefront, and continued advancements in human genomics will play a critical role, he says.
Forging Ahead
Let’s look forward to an exciting and productive 2015 that brings more cancer drugs to patients, more tools to prevent and detect cancer early, and more information to the cancer research community that works tirelessly to change the way we deal with this dreadful disease. We will be reporting on these advances next fall in our annual Cancer Progress Report.