Editors’ Picks, February 2026: FDA’s Accelerated Approval Pathway, Overcoming Resistance to KRAS Inhibition, and More
Olympic athletes are focused on accelerated movement, metabolism, and fighting (wind) resistance. So, too, are cancer researchers. This month’s Editors’ Picks include new insights into the impact of the accelerated drug approval pathway, the role of metabolism in prostate cancer progression, and ways to overcome resistance to KRAS inhibitors. And while the 2026 Olympic trials are a thing of the past, clinical trials sure aren’t, so be sure to check out the results from two clinical trials featured in February’s picks—one for head and neck cancer and another for breast cancer.
Read on for the abstracts of all the studies highlighted by the editors of the 10 peer-reviewed journals of the American Association for Cancer Research (AACR) and follow the provided links for free access to the full-text articles, available for a limited time.
Journal: Blood Cancer Discovery
Inhibition of the tyrosine kinase BTK is a major therapeutic strategy for treating B-cell malignancies, including chronic lymphocytic leukemia (CLL). However, resistance can emerge when tumor cells acquire mutations that abrogate drug binding or when BTK activates B-cell receptor (BCR) signaling by mechanisms independent of its kinase activity. In this study, we identified upregulation of PKCβ in samples from patients with CLL resistant to BTK inhibitors (BTKi) and characterized the PKCβ inhibitor MS-553. MS-553 reduced BCR and Wnt/β-catenin signaling, overcame stromal cell mediated protection, and synergized with venetoclax in CLL samples. MS-553 also retained the cytotoxicity and inhibition of both BCR and Wnt/β-catenin signaling in models (cell lines and primary samples) of covalent BTKi-resistant (C481S BTK) and noncovalent BTKi-resistant (T474I or L528W BTK) CLL. Furthermore, MS-553 delayed disease progression and prolonged survival in the Eμ-MTCP1 murine model of CLL. Collectively our results demonstrate that selective inhibition of PKCβ has the potential to overcome BTKi-resistant CLL.
Significance: This study identified and characterized PKCβ as a therapeutic target in CLL, including CLL resistant to BTKis. Inhibition of PKCβ suppressed both BCR and Wnt/β-catenin signaling, delayed disease progression in vivo, overcame BTKi resistance mechanisms, and enhanced response to BCL-2 inhibition.
A related commentary was published in the January issue, where this article was highlighted.
Journal: Cancer Discovery

Glioblastomas are incurable primary brain tumors that depend on neural-like cellular processes, tumor microtubes (TM), to invade the brain. TMs also interconnect single tumor cells to a communicating multicellular network that resists current therapies. In this study, we developed a combined, comprehensive in vitro/in vivo anti-TM drug screening approach, including machine learning–based analysis tools. Two protein kinase C (PKC) modulators robustly inhibited TM formation and pacemaker tumor cell–driven, TM-mediated glioblastoma cell network communication. As TM-unconnected tumor cells exhibited increased sensitivity to cytotoxic therapy, the PKC activator TPPB was combined with radiotherapy, and long-term intravital two-photon microscopy paired with spatially resolved multiomics revealed anti-TM and antitumor effects. TPPB treatment also decreased the expression of tweety family member 1 (TTYH1), a key driver of invasive TMs. Our study establishes a novel screening pipeline for anti-TM drug development, identifies a TM master regulator pathway, and supports the approach of TM targeting for efficient brain tumor therapies.
Significance: Cancers can hijack neural properties to grow, disseminate, and to resist therapies, but effective drug development pipelines against these features are missing. Here, we establish a compound screening approach that allowed the identification of PKC modulators that target cancer cell–intrinsic neurodevelopmental mechanisms, suggesting a new class of neuroscience-instructed cancer therapeutics.
This article was featured on the cover of the February issue. Learn more about this study at the upcoming AACR Annual Meeting 2026, where first author and 2026 NextGen Star Daniel D. Azorín, PhD, will present his research on this topic.
Journal: Cancer Epidemiology, Biomarkers & Prevention
Reconstructing the Origin and Demographic Expansion of the TP53 p.R337H Founder Variant in Brazil
Background: The TP53 p.R337H variant is a well-characterized founder mutation with an unusually high prevalence in Brazil, particularly in the South and Southeast regions, where it affects approximately one in 300 individuals. It is recurrently associated with pediatric adrenocortical tumors and malignancies within the Li–Fraumeni syndrome spectrum but exhibits markedly incomplete penetrance, suggesting the influence of additional genetic and/or environmental modifiers. Although TP53 p.R337H occurs at low frequencies in the Iberian Peninsula, its origin, timing, and dissemination pattern within Brazil have remained unresolved.
Methods: We integrated population–genetic inference with historical demographic modeling to reconstruct the introduction and expansion of the TP53 p.R337H in Brazil.
Results: Our analyses support a founder effect arising from a single founding event of European origin during the early Portuguese colonization period. The subsequent geographic dissemination of the variant parallels historical population growth, particularly in southern Brazil. The striking regional enrichment can be explained without invoking multiple introduction events or complex migratory scenarios.
Conclusions: These findings clarify the evolutionary and demographic history of TP53 p.R337H in Brazil and underscore its founder effect origin during European colonization.
Impact: This study highlights the value of integrating population genetics with historical data to elucidate the spread of medically relevant founder variants. Understanding the dissemination of TP53 p.R337H will enhance public health planning and genetic counseling strategies in Brazil.
A related commentary was published in the February issue.
Journal: Cancer Immunology Research
Tumor-draining lymph nodes are a pivotal site for antitumor T-cell priming. However, their mechanistic roles in cancer immune surveillance and immunotherapy response remain poorly defined. Intratumor (IT) virotherapy generates antitumor T-cell immunity through multifaceted engagement of innate antiviral inflammation. In this study, we identify type-I interferon (IFNI) signaling in glioma-draining cervical lymph nodes as a mediator of IT polio virotherapy. Transient IFNI signaling after IT administration was rescued by cervical perilymphatic infusion (CPLI) virotherapy, targeting cervical lymph nodes directly. Dual-site (IT plus CPLI) virotherapy induced profound inflammatory reprogramming of cervical lymph nodes, enhanced viral RNA replication and IFNI signaling in dendritic cells and high endothelial venules, augmented antiglioma efficacy in mice, and was associated with T-cell activation in patients with recurrent glioblastoma. A phase II clinical trial of IT plus CPLI polio virotherapy is ongoing (NCT06177964). This study implicates the lymphatic system as a virotherapy target and demonstrates that CPLI virotherapy has the potential to complement brain tumor immunotherapy.
A related commentary was published in the February issue.
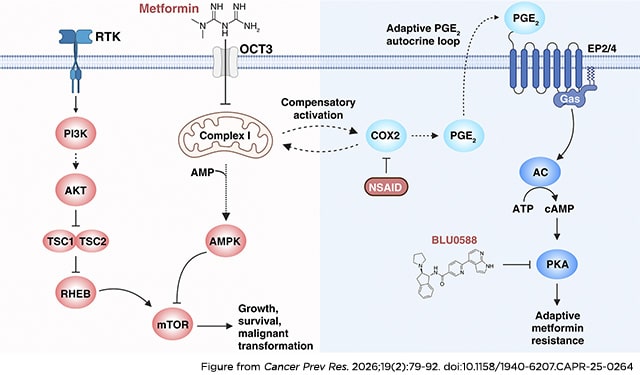
Journal: Cancer Prevention Research
Head and neck squamous cell carcinoma (HNSCC) is among the 10 most common cancers worldwide and is associated with high morbidity and poor survival. Diminished HNSCC outcomes are often related to delayed diagnosis and treatment of occult progression of premalignant lesions, underscoring the need for effective and low-risk chemoprevention strategies. In this regard, metformin has shown promising clinical activity for HNSCC prevention. In this study, we performed a genome-wide CRISPR/Cas9 screen of metformin-treated HNSCC cells and identified the activation of PKA signaling as the top resistance pathway. We show that metformin mediates PKA activation in HNSCC cells and that PKA inhibition, when combined with metformin treatment, synergistically inhibits HNSCC growth. We found that metformin-induced PKA activation is mediated by a prostaglandin E2 autocrine loop, which can be blocked using cyclooxygenase-2 (COX2) inhibitors. Importantly, COX2 inhibition using nonsteroidal anti-inflammatory drugs (NSAID) combined with metformin treatment synergistically inhibits HNSCC cell growth and prevents the progression of oral premalignant lesions into invasive HNSCC in a model of tobacco-driven oral carcinogenesis. Together, these findings demonstrate that metformin and NSAID combination therapy may represent a promising therapeutic strategy for HNSCC chemoprevention.
Prevention Relevance: Our findings reveal that using metformin for head and neck cancer chemoprevention leads to compensatory activation of a PKA-driven resistance mechanism that can be blocked by cotreatment with NSAIDs. These findings provide a rationale for combining metformin with NSAIDs as a precision head and neck cancer chemoprevention strategy.
This study was featured on the cover of the February issue.
Journal: Cancer Research (February 1 issue)
Estrogen Production in Pancreatic Cancer Shapes a Tumor-Suppressive Stromal Microenvironment
Pancreatic ductal adenocarcinoma (PDAC) is a leading cause of cancer-related death due to a lack of effective therapeutic interventions. PDAC tumors are highly fibrotic, and stromal abundance is hypothesized to contribute to patient outcomes. However, stromal fibroblasts can be tumor promoting and tumor restraining, which could explain the disappointing clinical results of stromal-targeting therapies. In this study, we observed that serum levels of stromal biomarkers associated with favorable outcomes were significantly higher in female patients with PDAC compared with male patients. This was supported by in silico estimates of stromal abundance across solid cancers, as well as magnetic resonance elastography and tissue staining, which revealed that female patients with PDAC had stiffer tumor tissue. Gene expression analysis revealed that estrogen signaling instructs a stromal fibroblast phenotype that is associated with relatively indolent molecular subtypes and a more favorable prognosis, which is maintained by stromal expression of C-type lectin CLEC3B. Remarkably, estrogens were detected intratumorally, and pancreatic cancer cells expressed key enzymes for estrogen synthesis. Estrogen production in PDAC was fueled by the catabolism of stroma-derived branched-chain amino acids, which ultimately resulted in the production of steroid hormone precursors. Together, these data reveal important hormone-driven factors that limit tumor progression by reprogramming the tumor microenvironment and provide leads for therapy development for PDAC.
Significance: Estrogen produced by pancreatic cancer cells alters the phenotype of cancer-associated fibroblasts to shape a tumor-suppressive microenvironment that hampers aggressive disease phenotypes, which could guide the development of stromal-targeted therapies.
A related commentary was published in the February 1 issue.
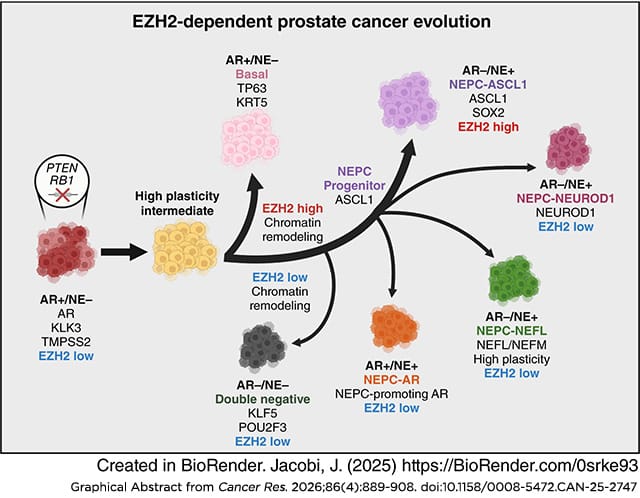
Journal: Cancer Research (February 15 issue)
Advanced prostate cancer remains a leading cause of cancer-related death among men due to disease progression in nearly all patients on standard-of-care therapy targeting the androgen receptor. An important mechanism driving therapeutic resistance is lineage plasticity, which enables prostate cancer cells to reprogram into lineage variants no longer dependent on androgen receptor signaling. As inhibitors of the histone methyltransferase enhancer of zeste homolog 2 (EZH2) are being evaluated clinically for the treatment of advanced prostate cancer, we investigated in this study how EZH2 affects prostate cancer lineage plasticity. Data from genetically engineered mice and human clinical samples demonstrated that genetic or pharmacologic suppression of EZH2 altered chromatin to expand active transcription factor programs. These changes in gene expression during prostate cancer progression increased the diversity of prostate cancer lineage variants that arose. EZH2 suppression did not inhibit disease progression nor therapeutic resistance in this context. These findings advance the current understanding of prostate cancer lineage plasticity and suggest that EZH2 inhibitors may be less effective in treating prostate cancer prone to lineage plasticity.
Significance: EZH2 suppression diversifies prostate cancer lineage plasticity, which has implications for EZH2-targeted therapies that are being evaluated for prostate cancer treatment.
A related commentary was published in the February 15 issue.
Journal: Clinical Cancer Research (February 1 issue)
Purpose: We report long-term survival and comprehensive molecular biomarker analyses of a phase II trial evaluating the combination of cetuximab and nivolumab in recurrent and/or metastatic (R/M) head and neck squamous cell carcinoma (HNSCC).
Patients and Methods: The long-term follow-up data were obtained from a phase II trial (NCT03370276). Archived tumors and serially collected plasma cell-free DNA were characterized by comprehensive genomic analyses. Immune markers were measured in tumor cores and margins by multiplex IHC.
Results: At a median follow-up of 47.4 months, the median overall survival (OS) and 2-year OS rate were 12.7 months and 32% in all evaluable patients (n = 88) and 17.5 months and 35% in patients who had no prior therapy for R/M HNSCC (n = 43). The survival efficacy was similar between patients with p16-negative and p16-positive tumors, but the response rate was significantly higher in patients with p16-negative tumors. The clonal tumor mutational burden, hypoxia score, and EGFR pathway score were significantly higher in p16-negative compared with p16-positive tumors. Loss of heterozygosity of MHC class I was significantly more frequent in nonresponders, and APOBEC-associated mutagenesis was elevated in responders. Gene expression profiling and multiplex IHC analyses revealed a more inflamed tumor microenvironment in responders regardless of p16 status.
Conclusions: Our long-term follow-up study indicates that the combination of cetuximab and nivolumab is efficacious and tolerable and that patients with p16-negative R/M HNSCC may have greater benefit from this combination.
This study was featured on the cover of the February 1 issue.
Journal: Clinical Cancer Research (February 15 issue)
Purpose: Based on the results from SOLTI-PATRICIA trial (NCT02448420) cohorts A/B, a direct comparison with standard-of-care treatments is needed to evaluate the efficacy and safety of palbociclib, trastuzumab, and endocrine therapy (ET) in PAM50 luminal A/B pretreated patients.
Patients and Methods: SOLTI-PATRICIA cohort C is a randomized, multicenter, prospective, open-label, phase II study. Pretreated patients with HER2-positive, hormone receptor–positive, and PAM50 luminal A/B advanced breast cancer were randomized 1:1 to receive either i) the triplet regimen or ii) trastuzumab-based treatment of physician’s choice (TPC). Patients allocated in the TPC arm were eligible for re-randomization upon disease progression, if the inclusion criteria were still met. The primary endpoint was investigator-assessed progression-free survival (PFS) per RECIST version 1.1.
Results: A total of 264 participants were prescreened from August 2019 to 2023, and 73 patients were randomly assigned (including seven re-randomizations). In the TPC arm, 48.5% were treated with trastuzumab plus chemotherapy, 39.4% with trastuzumab emtansine, and 12.1% with trastuzumab plus ET. The triplet was associated with a significantly better PFS compared with TPC [stratified hazard ratio = 0.52; 95% confidence interval (CI), 0.29–0.95; two-sided P = 0.03]. PFS rates after 24 months were 24.0% with the triplet and 4.3% in the TPC arm. The overall response rate was 18.9% (95% CI, 8.6–35.7) and 7.1% (95% CI, 1.2–25.0), respectively. In the triplet arm, grade ≥3 adverse events occurred in 61.5% of patients, with neutropenia being the most frequent (53.9%). No permanent discontinuations due to toxicity were observed.
Conclusions: Combining palbociclib, trastuzumab, and ET was safe and significantly improved PFS, compared with TPC, in previously treated patients with HER2-positive, PAM50 luminal A/B advanced breast cancer.
Journal: Molecular Cancer Research
Bone Metastatic Progression of Prostate Cancer Is Regulated by TRIM28–LDHA-Mediated Metabolism
Castration-resistant prostate cancer, an advanced stage of prostate cancer, often leads to fatal bone metastasis. The vast majority of patients with prostate cancer who present with bone metastases suffer from bone lesions and other complications. Androgen receptor inhibitors, although improved, lack curative efficacy, necessitating an urgent demand for the development of innovative therapeutic strategies. TRIM28, also known as Krüppel-associated box–associated protein 1, is a transcription factor regulated by site-specific phosphorylation. Our recent study demonstrated that p90 ribosomal S6 kinase 1 is the protein kinase that directly phosphorylates TRIM28 at S473; as such, pS473-TRIM28 promotes the transcriptional activation of its gene targets. In this study, we reveal that TRIM28-S473 phosphorylation is readily detected in castration-resistant prostate cancer bone metastases, which is consistent with the previous report that p90 ribosomal S6 kinase is activated in prostate cancer bone metastases. Using bioinformatic and genomic analyses, we uncovered that lactate dehydrogenase A (LDHA) is a novel TRIM28-induced gene in bone metastatic prostate cancer. TRIM28 promotes the transcriptional activation of LDHA in a pS473-TRIM28–dependent manner. As such, TRIM28 is involved in LDH-related activities including lactate production and glycolysis. We also demonstrate that the TRIM28–LDHA axis is required for prostate tumor progression using an orthotopic bone injection model. Lastly, the application of an LDH inhibitor mitigates prostate cancer development in the bone. In summary, our study reveals an important role of the TRIM28–LDHA axis in prostate cancer progression in the bone, which may be targeted to mitigate the disease in the metastasis stage.
Implications: TRIM28 upregulates LDHA and glycolysis, propelling prostate tumors in the bone; pharmacologic LDH blockade mitigates disease.
Journal: Molecular Cancer Therapeutics
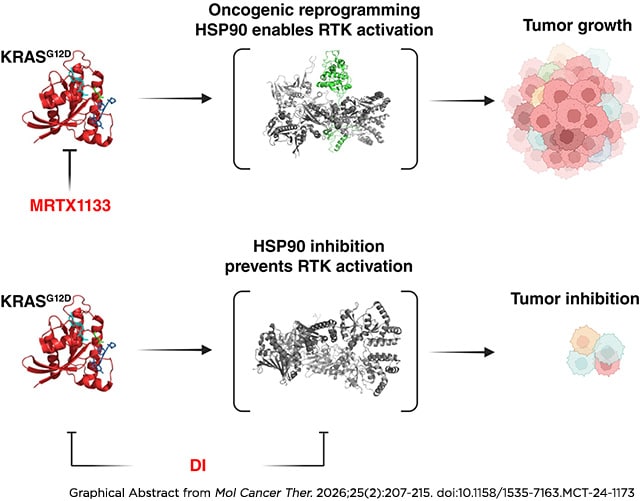
Dual Inhibitors of KRASG12D and HSP90 Are Effective against KRASG12D Inhibitor Resistance
KRASG12D is a common oncogenic driver mutation in diverse cancers, including non–small cell lung cancer, colorectal cancer, and pancreatic cancer. KRASG12D inhibitors have recently progressed into clinical trials but will likely face innate or acquired drug resistance similar to that which has been observed for KRASG12C inhibitors, such as activation of receptor tyrosine kinases, KRAS independence, and reactivation of RAS–MAPK signaling. This study investigates heterobifunctional small-molecule dual inhibitors that simultaneously target both KRASG12D and protein chaperone HSP90 in KRASG12D-mutated cancer cell lines and patient-derived organoids. Our findings reveal that the efficacy of the clinical-stage KRASG12D inhibitor MRTX1133 varies, with notable resistance being observed in some cell line and organoid models. In contrast, KRASG12D–HSP90 dual inhibitors were found to broadly display superior effectiveness in inducing apoptosis, reducing cell viability, and suppressing key downstream signaling pathways such as AKT and ERK1/2 in MRTX1133-resistant models. The rationale for targeting HSP90, which is preferentially activated in cancer cells, alongside KRASG12D, arises from the ability of HSP90 inhibition to destabilize substrate client proteins that are essential for cancer cell survival and have also been implicated in resistance to KRAS inhibitors. This dual-inhibitor approach presents a promising new strategy to combat de novo and acquired drug resistance in KRASG12D-mutated cancers and potentially paves the way for improved clinical outcomes by addressing the complex molecular mechanisms underlying cancer cell evolution that enables resistance to conventional inhibitors.
This study was featured on the cover of the February issue.
Journal: Cancer Research Communications
Real-World Clinical Oncology Outcomes Associated with the Accelerated Approval Pathway
The accelerated approval (AA) pathway expedites treatment access for patients with unmet medical needs. However, evidence on the program’s impact is lacking. This study assessed real-world clinical outcomes for AA drugs in solid tumor cancers. A nationwide electronic health record (EHR)-derived deidentified database was utilized to explore the impact of the AA pathway in patients with selected solid tumors from 2011 to 2023. Included patients received AA or non-AA products with the same indication. Outcomes were real-world progression-free survival (rwPFS) and overall survival (OS). Population-level health impacts were estimated via market share and real-world progression-free life years (rwPFLYs) and life years (LYs). Twenty-three AA indications were studied. At study completion, 83% of indications had their clinical benefit verified, whereas 17% had been withdrawn from the market. Overall, 78% of indications demonstrated significant improvements in rwPFS or OS, whereas none reported significantly worse rwPFS versus controls. In total, 44% and 48% of indications had >30% rwPFS and OS improvement, respectively. More indications with sample sizes >100 in the AA cohort demonstrated significant rwPFS gains [80% (12/15)] versus cohorts <100 [38% (3/8)]. An estimated 118,107 patients received AA drugs during their AA window, resulting in 56,399 rwPFLYs and 50,848 LYs gained. Most solid tumor AA indications were associated with improved clinical outcomes, providing substantial US population-level health gains. This EHR analysis provides insight into whether the AA pathway appropriately targets promising beneficial therapies. Expanding beyond randomized controlled trial evidence and conventional primary endpoints may be necessary to verify clinical benefit, particularly in rare tumors.
Significance: This study (the first examining the real-world outcomes associated with the AA period) found improved clinical outcomes for most AA drugs studied compared with existing treatments in solid tumor cancers. The improvements observed suggest potential health improvements at the US population level and the possibility of the need to look beyond randomized controlled evidence and traditional primary endpoints to verify the clinical benefit of AA drugs for relatively rare conditions.
To learn more about this topic, check out our blog post about another study that examined the fate of drugs approved through the accelerated approval pathway.