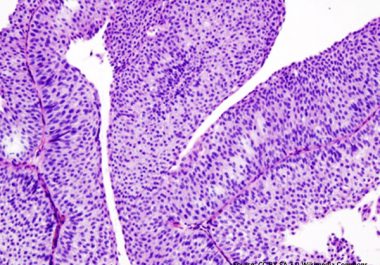
Targeting Bladder Cancer
A molecularly targeted therapeutic was approved by the FDA to treating certain patients with urothelial carcinoma, the most common form of bladder cancer.


A molecularly targeted therapeutic was approved by the FDA to treating certain patients with urothelial carcinoma, the most common form of bladder cancer.
A large study of postmenopausal women showed strong benefit from smoking cessation in lowering the risk of bladder cancer. Bladder cancer is the most common malignancy of the urinary system. Some 80,470 new diagnoses...
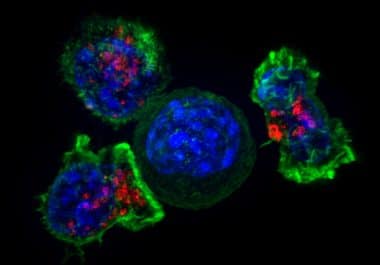
The FDA’s recent announcement means the immune checkpoint inhibitor pembrolizumab is now approved for five forms of cancer. The U.S. Food and Drug Administration (FDA) recently approved pembrolizumab (Keytruda) for the treatment of certain...
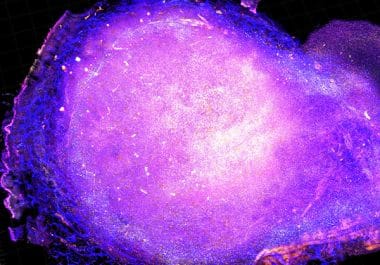
The FDA approved an immune checkpoint inhibitor – a type of immunotherapy – to treat certain patients with bladder cancer.
The FDA has approved a new immune checkpoint inhibitor, durvalumab, for the treatment of certain bladder cancer patients.
An immune checkpoint inhibitor – a form of immunotherapy – has had its use expanded for use in the treatment of bladder cancer.

The May 2016 approval of atezolizumab provided the first new treatment in more than 30 years for patients with the most common type of bladder cancer.