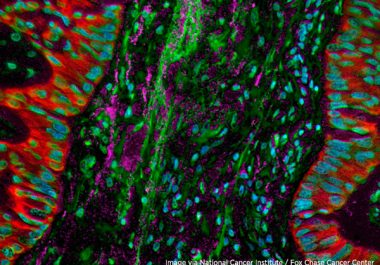
Targeting Non-Small Cell Lung Cancer
The FDA approved a new molecularly targeted therapeutic for certain patients with lung cancer driven by a mutation in the MET gene.


The FDA approved a new molecularly targeted therapeutic for certain patients with lung cancer driven by a mutation in the MET gene.
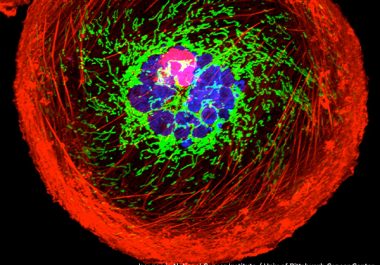
The FDA approved a type of molecularly targeted therapeutic called an antibody-drug conjugate for the treatment of metastatic triple-negative breast cancer.
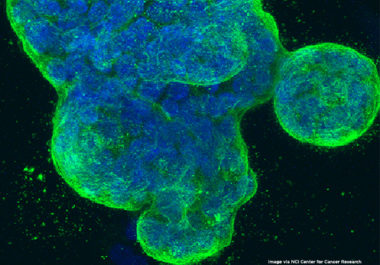
The FDA has approved a new molecularly targeted therapeutic for treating certain patients with advanced or metastatic HER2-positive breast cancer.
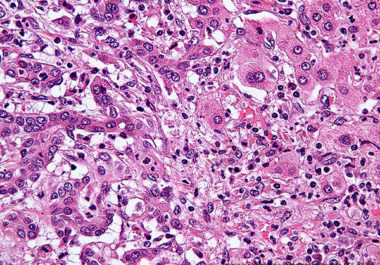
The first molecularly targeted therapeutic for use in the treatment of bile duct cancer has been approved by the FDA.
Treating a rare cancer with a chemotherapy gel.

The FDA has approved a molecularly targeted therapeutic for treating certain patients with neurofibromatosis type 1 who have inoperable tumors.
The FDA has approved a new combination of molecularly targeted therapeutics for treating metastatic colorectal cancer with a BRAF V600E gene mutation.
The FDA has approved a new combination of immunotherapy and chemotherapy as an initial treatment for extensive-stage small cell lung cancer.
The FDA approved using a combination of nivolumab and ipilimumab, two immune checkpoint inhibitors, to treat certain patients with hepatocellular carcinoma.
The FDA has approved a new therapeutic to treat adults with multiple myeloma whose cancer has progressed despite receiving at least two other treatments.