New Type of Cancer Immunotherapeutic Approved by FDA: What It Means for Leukemia Patients
Earlier this month we witnessed another leap forward for immunotherapy when, more than five months earlier than expected, the U.S. Food and Drug Administration (FDA) approved blinatumomab (Blincyto) for the treatment of certain patients with acute lymphoblastic leukemia (ALL).
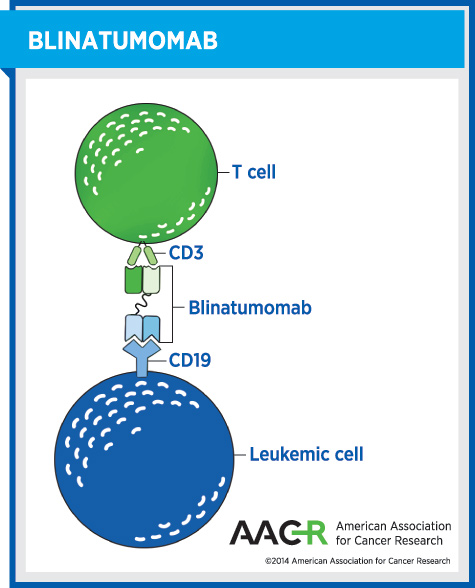
Blinatumomab is the first in a new class of cancer immunotherapeutics called bispecific T cell-engager (BiTE) antibodies. These agents function as a connector, bringing immune cells called killer T cells into close proximity with cancer cells, which are then eliminated by the killer T cells. In the case of blinatumomab, it attaches on one hand to a molecule called CD3 on killer T cells and on the other to CD19, a molecule found on the surface of most B-cell ALL cells. By attaching to these molecules on the different cells, blinatumomab brings the two cell types together, directing the killer T cells to home in on the leukemia cells.
The patients who will benefit from the FDA approval are adults with relapsed or refractory precursor B-cell ALL without the Philadelphia chromosome. Although this is a rare form of cancer—fewer than 2,250 U.S. adults are projected to receive a precursor B-cell ALL diagnosis in 2014—patients with this aggressive disease have a dismal outlook, with a median survival of between two and eight months, and there is an urgent need for new treatment options.
The approval of blinatumomab was based on initial results from a phase II clinical trial that showed that after two cycles of treatment with the cancer immunotherapeutic, 32 percent of participants achieved complete remission—meaning that 5 percent or fewer cells in the patient’s bone marrow were leukemia cells, and that the patient had no evidence of disease and full recovery of blood counts. These responses lasted for a median of 6.7 months.
Given that the approval centered on complete remission data, blinatumomab’s manufacturer, Amgen, is required by the FDA to conduct a study to confirm that the cancer immunotherapeutic improves survival for patients.
In addition to being on the surface of precursor B-cell ALL cells, CD19 is found on a number of types of B-cell lymphoma cells, and blinatumomab is being investigated as a potential treatment for these diseases. CD19 is also the target of other promising investigational cancer immunotherapeutics, most notably CTL019 and JCAR015. These two treatments are referred to as chimeric antigen receptor (CAR) T-cell therapies, and after reports of encouraging clinical responses, both received breakthrough therapy designation from the FDA this year for the treatment of relapsed or refractory precursor B-cell ALL. With this designation intended to enhance the path to regulatory approval, it is hoped that further groundbreaking advances in immunotherapy occur for patients with CD19-positive leukemia or lymphoma in the near future.