AACR Virtual Annual Meeting II: Insights into Cancer Metastasis Reveal Potential Therapeutic Strategies
The prognosis for a patient with cancer depends on several factors, including the cancer’s stage. Patients whose cancers are diagnosed at earlier stages, when the cancer is localized to the organ in which it originated, tend to have better survival rates than patients who are diagnosed after the cancer has spread—or metastasized—to other parts of the body.
Metastasis is a multi-step process that requires cancer cells to migrate from the primary tumor, invade blood or lymphatic vessels (intravasation), travel to other regions of the body, exit the blood or lymphatic vessel into the metastatic site (extravasation), and colonize and proliferate at the metastatic site. While these steps are understood to be important for metastasis, the mechanisms underlying each step remain unclear for many cancers.
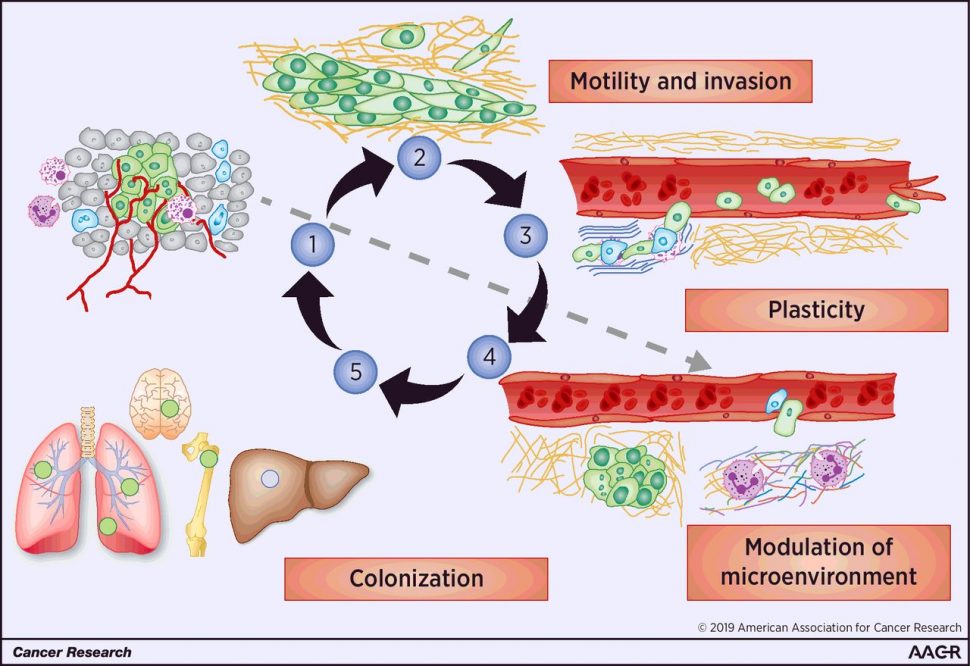
Understanding how metastasis develops may reveal new therapeutic targets to help prevent the spread of cancer and improve survival rates. A minisymposium at the AACR Virtual Annual Meeting II, which was held online June 22-24, featured several studies that revealed new insights into metastatic mechanisms.
Cisplatin promotes lung cancer metastasis
Despite being an effective treatment for primary tumors, chemotherapy is often not as effective for metastatic cancers, explained Giulia Bertolini, PhD, from the Fondazione IRCCS Istituto Nazionale dei Tumori in Milan. She explained that this may be due to tumor-promoting processes activated by chemokines, which are released upon chemotherapy-induced cellular damage.
Previous work from Bertolini and colleagues found that cisplatin, a platinum-based chemotherapy, led to the selection of CD133 and CXCR4-positive chemoresistant cancer stem cells, which are associated with metastasis in lung cancer. Furthermore, they found that the inhibition of the chemokine receptor CXCR4 prevented such selection. These findings led Bertolini and colleagues to explore the role of CXCR4 and its ligand SDF-1 on the pro-metastatic effects induced by cisplatin.
In her presentation, Bertolini reported that cisplatin treatment led to an increase in the number of CXCR4-positive inflammatory monocytes and promoted the release of SDF-1 in the lung tissue of tumor-free mice. These effects were not observed when cisplatin treatment was accompanied by a CXCR4 inhibitor nor when mice were treated with other common chemotherapy agents, suggesting that the effects were dependent on CXCR4 and specific to cisplatin treatment. The researchers also found increased metastasis and metastasis-associated macrophages in mice that were injected with cisplatin-treated human lung cancer cells, but not in those injected with cells treated with cisplatin and the CXCR4 inhibitor. Similarly, there was an increase in the number of metastasis-initiating cells in xenograft models treated with cisplatin, but not in those also treated with the inhibitor.
By examining pre- and post-treatment tumor biopsy samples from patients with stage III non-small cell lung cancer, Bertolini and colleagues found that neoadjuvant cisplatin treatment led to a significant increase in SDF-1 levels, which was correlated with reduced disease-free and overall survival.
Bertolini concluded that cisplatin treatment may promote metastasis by activating the SDF-1/CXCR4 axis, which leads to the recruitment of CXCR4-positive inflammatory monocytes and the expansion of CXCR4-positive metastasis-initiating cells. She added that including a CXCR4 inhibitor could potentially improve responses to cisplatin treatment.
Cell state diversity promotes metastasis in melanoma
Another study presented during the session examined the contributions of different melanoma cell states on metastasis. As explained by Nathaniel Campbell, a student in the Weill Cornell/Rockefeller/Sloan-Kettering Tri-institutional MD-PhD Program, melanomas exhibit intratumor heterogeneity due to the presence of both invasive and proliferative cell states. These states are dynamic, and switching from one cell state to the other is referred to as “phenotype switching.”
Campbell and colleagues utilized a zebrafish model of melanoma to explore how each cell state contributes to metastasis. Using fluorescence microscopy to observe the spread of each cell type, they found that invasive cells were more metastatic than proliferative cells. Furthermore, gene expression analyses revealed that invasive cells expressed a cell-cell adhesion gene signature; consistent with this gene signature, the invasive cells were found to form larger clusters than the proliferative cells. Campbell and colleagues also observed that invasive and proliferative cells clustered with each other, and that proliferative cells formed more metastases when clustered with invasive cells than when they were alone.
Gene expression analyses of both cell states revealed that members of the transcription factor AP-2 family (TFAP) were highly expressed in proliferative cells, and that deletion of TFAP genes drove cells toward a more invasive state with slower proliferation, greater extravasation, and greater clustering.
Campbell concluded that proliferative and invasive melanoma cells cooperate to extravasate and promote metastasis. He suggested that the clustering of proliferative cells with the more metastatic invasive cells might allow secondary tumors to maintain the heterogeneity of the primary tumor without the need for rapid phenotype switching at the metastatic site. Furthermore, he hypothesized that the heterogeneous clusters could be therapeutic targets.
Inhibition of the HIPPO pathway suppresses glioblastoma growth and migration
Recurrence of the lethal brain cancer glioblastoma is driven by migratory and invasive cells that generate secondary tumors, ultimately killing the patient. Anne Marie Barrette, PhD, currently a postdoctoral researcher at Stanford University, presented her graduate thesis research on glioblastoma cell migration and therapeutic interventions, which was conducted at the Icahn School of Medicine at Mount Sinai.
Barrette and colleagues determined that the TEAD family of transcription factors were highly expressed in glioblastoma cells. TEAD is a member of the HIPPO signaling pathway, which has been implicated in tissue growth, cellular migration, proliferation, and organ development. TEAD transcriptional activity is regulated by the YAP and TAZ proteins, which translocate to the nucleus and bind TEAD to stimulate its activity.
Barrette and colleagues explored the potential of verteporfin, a drug that prevents nuclear translocation of YAP/TAZ, to inhibit TEAD activity and suppress glioblastoma migration. Verteporfin is approved by the U.S. Food and Drug Administration for the treatment of macular degeneration and has previously been shown to suppress migration and proliferation of ovarian and pancreatic cancer cells.
The researchers confirmed that verteporfin downregulated HIPPO pathway members and known TEAD target genes in four glioblastoma cell lines. Gene expression analyses of verteporfin-treated cells revealed that downregulated genes included those involved in cell motility, migration, and cell adhesion. Furthermore, verteporfin treatment reduced cellular migration in multiple in vitro migration assays, raising the possibility that verteporfin could be used to suppress migration of glioblastoma cells in vivo.
To understand its therapeutic potential, Barrette and colleagues examined the impact of verteporfin on glioblastoma migration in mouse models. They found that verteporfin was able to penetrate the brain and that it downregulated HIPPO pathway members and reduced the amount of YAP in brain cell nuclei. Furthermore, verteporfin suppressed the growth and migration of transplanted patient-derived glioblastoma cells. In mice transplanted with patient-derived cells from primary glioblastomas, verteporfin treatment alone significantly improved survival compared to vehicle-treated mice. Mice that were transplanted with recurrent glioblastoma cells showed no improvement in survival when treated with either verteporfin alone or the current standard of care (temozolomide and radiation), but they did have significant improvement in survival when treated with verteporfin in combination with temozolomide and radiation.
Barrette concluded that verteporfin downregulates TEAD target genes, suppresses migration of glioblastoma cells, and confers a survival benefit alone or in combination with the current standard of care.
Together, these three studies demonstrate some of the myriad mechanisms underlying cancer metastasis and illustrate how understanding such mechanisms can reveal potential therapeutic targets that could ultimately improve survival for patients with cancer.



