Taking a BiTE Out of Cancer
Our immune systems are equipped to recognize and destroy cancer cells—if they can find them, that is. And with tens of trillions of cells in the human body and the masterful hiding skills of cancers, that’s a big if.
But what if we could step in and show immune cells where the cancer is?
That’s the idea behind bispecific T-cell engagers, or BiTEs for short. Comprised of a two-headed antibody, they can simultaneously bind one protein on a cancer cell and another on a type of immune cell known as a T cell, bringing the two cells in proximity to one another.
The idea for such a strategy was proposed in the 1960s by Alfred Nisonoff and colleagues, who showed that it was possible to engineer an antibody against two different proteins and bring together two different cell types.
Now, more than six decades later, seven BiTEs are approved to treat cancer, and several more are in clinical testing. In this post, we’ll provide an overview of this emerging class of therapies and discuss where current research is headed.
What does a BiTE look like?
A typical antibody is shaped like the letter ‘Y’, with both arms of the ‘Y’ binding one target protein (or “antigen”) and the stem of the ‘Y’ activating immune signaling. To make a BiTE, an arm fragment from the ‘Y’ portion of an antibody targeting one antigen is joined to an arm fragment from the ‘Y’ portion of an antibody targeting another antigen, resulting in a molecule that has two different antigen-binding sites. BiTEs lack the stem region of a typical antibody; instead, the arms are joined by a flexible linker.
Each arm of the BiTE targets a distinct protein—one arm is engineered to bind an antigen on the cancer cell (ideally one that is not expressed on normal cells), and the other arm is designed to bind a protein called CD3 on the surface of T cells (see Figure).
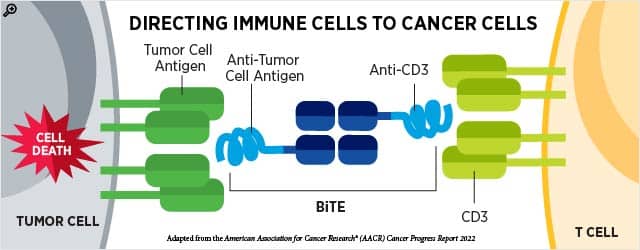
The T cell found the cancer cell—now what?
To avoid nonspecific activity, the BiTE must bind to both the cancer cell and the T cell to stimulate the T cell’s antitumor functions. Once this happens, signaling pathways in the T cell are activated, resulting in the release of various molecules from the T cell. The released molecules serve multiple purposes: 1) some poke holes in the cancer cell, forcing the cancer cell to self-destruct, 2) others activate cell death pathways within the cancer cell, and 3) some prompt surrounding cancer cells to display the target protein on their surfaces, so they, too, can be targeted by the BiTE. Together, these events constitute a multipronged attack on cancer cells.
What types of cancers can be treated with BiTEs?
Currently, BiTEs are FDA-approved to treat patients with certain blood cancers, including multiple myeloma, leukemia, and lymphoma. The approved BiTEs target proteins commonly expressed by malignant blood cells, including B-cell maturation antigen (BCMA), CD20, CD19, and GPRC5D.
- Teclistamab (Tecvayli) is approved for certain patients with relapsed or refractory multiple myeloma.
- Elranatamab (Elrexfio) is approved for certain cases of heavily pretreated multiple myeloma.
- Talquetamab (Talvey) is approved for certain patients with heavily pretreated multiple myeloma.
- Blinatumomab (Blincyto) is approved to treat adult and pediatric patients with certain B cell-derived acute lymphoblastic leukemias.
- Glofitamab (Columvi) is approved for select patients with certain types of pretreated diffuse large B-cell lymphomas (DLBCL) or large B-cell lymphomas (LBCL).
- Epcoritamab (Epkinly) is approved for select patients with certain types of pretreated DLBCL or LBCL.
- Mosunetuzumab (Lunsumio) is approved for certain patients with relapsed or refractory follicular lymphoma.
What are some challenges to BiTE therapy?
Despite the success of BiTEs for some patients, these therapeutics come with the risk of life-threatening toxicities, including neurological toxicities and cytokine release syndrome caused by immune overactivation. BiTEs can also cause depletion of the patient’s immune cells, which leaves them vulnerable to severe bacterial or viral infections.
Clinicians utilize various tools to manage these issues, including steroids to treat neurological toxicities and immune-suppressing drugs with or without steroids for patients with cytokine release syndrome. To mitigate the risk of infections, clinicians sometimes administer antibiotics prophylactically. Another strategy is to boost the patient’s immune system by delivering donor antibodies during BiTE therapy. The potential of this approach for preventing serious infections was demonstrated in a recent study published in Blood Cancer Discovery, a journal of the AACR; however, the results still need to be confirmed in larger studies.
Another challenge is the development of treatment resistance. Cancer cells may stop expressing the target antigen to evade detection by the BiTE, or they might increase the expression of certain immune-suppressing proteins to block the engaged T cells from being activated.
And while BiTEs have demonstrated dramatic efficacy for multiple blood cancers, they have been unsuccessful against solid tumors so far. Part of the reason for this is that many antigens expressed on the surface of solid tumors are also found on healthy cells throughout the body, which increases the likelihood that the BiTE will kill noncancer cells and potentially damage vital organs.
What’s next?
Researchers are exploring new iterations of the BiTE design in an effort to overcome some of the obstacles described above. Some designs under investigation incorporate an immune checkpoint inhibitor as part of the BiTE to prevent cancer cells from suppressing T-cell activation. Others target multiple tumor antigens at once, which can help keep the BiTE functional even if the cancer stops expressing one of the antigens.
And while BiTEs have so far been unsuccessful against solid tumors, related molecules called ImmTACs (for immune-mobilizing monoclonal T-cell receptors against cancer) have shown promise, with one such therapeutic, tebentafusp (Kimmtrak), approved to treat uveal melanoma.
Like BiTEs, ImmTACs simultaneously bind proteins on cancer cells and T cells to bring the two cell types together. But unlike BiTEs, ImmTACs use a T-cell receptor instead of an antibody fragment to recognize proteins on cancer cells, thereby allowing these molecules to bind intracellular proteins that have been processed and presented externally instead of being restricted to proteins expressed on the cell surface. This is an important difference, as most cancer-specific proteins in solid tumors are expressed inside the cell.
At the recent AACR-NCI-EORTC International Conference on Molecular Targets and Cancer Therapeutics, researchers discussed strategies to improve ImmTACs for solid tumors. One approach is to engineer ImmTACs to have a low affinity to the T-cell protein CD3. The rationale behind this design is that weaker CD3 binding may prevent overactivation of the immune response and could, therefore, allow the drug to be administered at higher doses without increasing the risk for cytokine release syndrome.
In preclinical testing, this approach was shown to be effective at killing tumor cells while generating fewer cytokines, indicating that it might be less likely to cause cytokine release syndrome. This investigational ImmTAC, GEN1047, is designed to target the B7H4 immune checkpoint protein on cancer cells; it showed promising preclinical activity in ovarian cancer models and has entered clinical testing, according to the presenter.
IMA402 is another investigational ImmTAC with low CD3 affinity. IMA402 is designed to target the PRAME protein and has demonstrated clinical activity against several solid tumor types, including lung, ovarian, uterine, skin, and synovial cancers, another researcher reported.
In the ongoing game of hide and seek, cancer cells too often have the advantage, but BiTEs and ImmTACs constitute a promising class of therapeutics that could give cancer-fighting immune cells an edge. By bridging cancer cells and immune cells, these molecules are helping us tap into the incredible antitumor potential of the immune system.