
AACR Blog Turns 3: Check Out Our Top 10 Posts
Three years ago, the American Association for Cancer Research (AACR) launched this blog with a welcome post from Chief...


Three years ago, the American Association for Cancer Research (AACR) launched this blog with a welcome post from Chief...

At the end of last week, the U.S. Food and Drug Administration (FDA) approved expanding the use of pembrolizumab...

Earlier this week, the U.S. Food and Drug Administration (FDA) announced that it had approved a new immune checkpoint...

On March 23, the U.S. Food and Drug Administration announced the first-ever approval of a treatment for patients with...
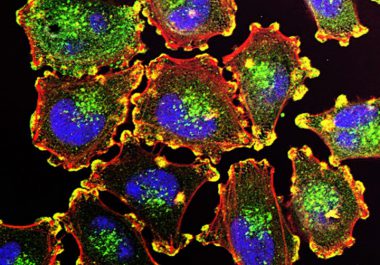
Identifying ways to increase the proportion of patients with melanoma who have clinically meaningful and durable responses to immune...
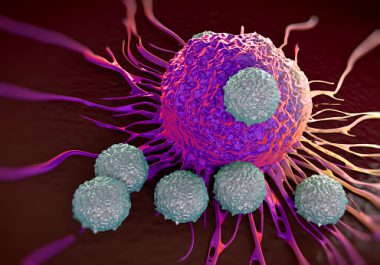
Now that a plethora of clinical trials have established positive responses from immunotherapies—immune checkpoint inhibitors, in particular—in patients with...

The U.S. Food and Drug Administration (FDA) has approved expanding the use of the immunotherapeutic pembrolizumab (Keytruda) to include...
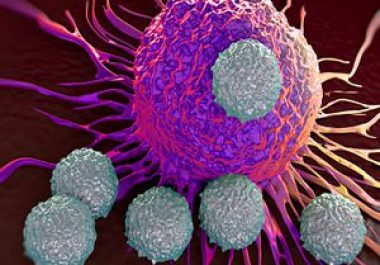
A hot topic in the field of cancer immunotherapy is: How do some cancers that initially respond to immune...

A look at the 10 most-read posts of the year on Cancer Research Catalyst illustrates the steady pace of...

The potential for developing new immunotherapies and expanding the use of existing ones will be the focus of the...