AACR Associate Members Receive Warm Reception at Early-career Hill Day
Heather K. Beasley was among the researchers who visited Washington, D.C to push for sustained NIH funding during AACR...

Heather K. Beasley was among the researchers who visited Washington, D.C to push for sustained NIH funding during AACR...

Yesterday marked a much anticipated milestone for the oncology community: The U.S. Food and Drug Administration (FDA) announced the...
A pair of studies published recently in journals of the American Association for Cancer Research bring to our attention...

Earlier this year, a study published in Cancer Discovery, a journal of the American Association for Cancer Research (AACR),...

A major objective of former Vice President Joe Biden’s National Cancer Moonshot Initiative is breaking down silos and sharing...

This week, the U.S. Food and Drug Administration (FDA) added two new treatments to the armamentarium for hematologic oncologists...

Patient enrollment in clinical trials has long been on the minds of oncologists and researchers. In 1990, fewer than...
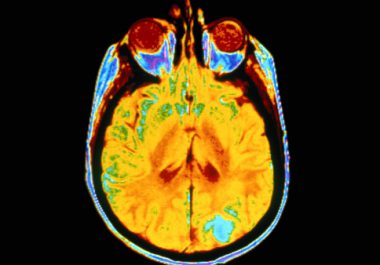
As we learned recently, Senator John McCain was diagnosed with glioblastoma multiforme, an aggressive form of central nervous system...

Yesterday, the American Association for Cancer Research (AACR) joined the American Cancer Society (ACS), the American Society of Clinical...

As President of the American Association for Cancer Research (AACR), Michael A. Caligiuri, MD, is a strong advocate for...

Receiving a cancer diagnosis often means navigating numbers, such as estimates of the chance of recurrence. But cancer patients...