The More You Know About Cancer Prevention
The World Health Organization estimates that 30% to 50% of all cancer cases are preventable.
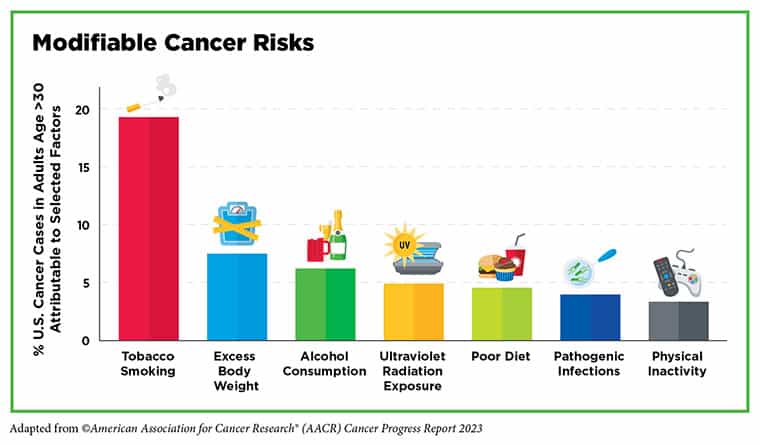
Yes, much of that can be attributed to healthy lifestyle choices, including physical activity, better nutrition, lack of smoking, and light to moderate alcohol consumption, but knowledge can also be a powerful cancer prevention tool. According to the American Association for Cancer Research (AACR) Cancer Progress Report 2023, the United States experienced a 33% decline in overall cancer mortality between 1991 and 2020 largely thanks to public health campaigns and policy initiatives implemented to reduce smoking and increase early detection of cancers, based on study findings in CA: A Cancer Journal for Clinicians.
As they say (or at least a series of public service announcements did in the ’90s), “The More You Know.” And in this case, the more you know about your cancer risks can be lifesaving, and what you don’t know can have negative consequences. A series of recent studies have found that some people may lack a certain level of clarity and/or awareness about some cancer prevention techniques such as screening and vaccination. This may cause gaps in ensuring benefit from available methods that can help detect cancer much earlier or outright prevent it.
Some Stool Samples Aren’t Fit to be Tested
One of the several initial screening options for colorectal cancer (CRC) is a self-administered fecal immunochemical test (FIT) in which an individual provides a stool sample to be examined for hidden blood. The FIT is either administered at a health care office (often with verbal instructions) or via a mail-order program (with written and/or info-graphical instructions). But 1 in 10 FITs could not be processed due to unsatisfactory samples, according to results from a study published in Cancer Epidemiology, Biomarkers & Prevention, a journal of the AACR.
At least part of the issue is individuals don’t fully comprehend what is required to provide a sample that is deemed satisfactory for testing, according to Rasmi Nair, MBBS, PhD, co-first author of the paper and an assistant professor at the Peter O’Donnell Jr. School of Public Health of UT Southwestern Medical Center. That was especially true for the mail-order program, which was 2.66 times more likely to produce unsatisfactory results.
Nair and her colleagues examined electronic health record (EHR) data of 56,980 individuals aged 50 to 74 who underwent FIT screening between 2010 and 2019 within the Dallas-based Parkland Health system. Parkland, which is considered a safety-net hospital, provides care to more than one million low-income, uninsured Dallas County residents. Overall, of the 10.2% FITs considered unsatisfactory, 51% were due to an inadequate specimen, 27% were attributed to incomplete labeling, 13% of the stool specimens were too old, and 8% had a broken or leaking container. Additionally, only 40.7% of individuals with unsatisfactory tests received follow-up FIT or colonoscopy screening within 15 months of the failed test.
Nair suggested that minimizing language and health literacy barriers could help, and the study authors pointed to visual instructions that showed positive results in improving sample collection in other studies. The authors also suggested that testing facilities include previously affixed patient labels or barcodes to minimize labeling errors as well as policy changes to allow using the sample ordering, mailing, or receiving date as the collection date—if the date is missing on the label itself and the sample is sent within the two-week widow. Finally, the authors also want to see a better system put in place to ensure proper follow-up.
“The fact that, in most instances, unsatisfactory FIT was not followed by a timely subsequent test highlights the need for systems to have a better, more comprehensive approach to tagging and following up unsatisfactory FIT,” said co-first author Po-Hong Liu, MD, a gastroenterology fellow at UT Southwestern Medical Center.
The Need for More HPV Vaccine Awareness
The vaccine for human papillomavirus (HPV) has shown tremendous results in preventing cervical cancer. In fact, a recent study examining cervical cancer cases in Scotland found zero cases among women born between 1988-1996 who were fully vaccinated against HPV between the ages of 12 and 13, according to a paper published in the Journal of the National Cancer Institute.
The HPV vaccine, however, can benefit men and protect against other cancers as well, including anal, oral, and penile cancers. But this fact may not be properly presented to Hispanic and Latino men who identify as sexual minorities, according to results presented at the 16th AACR Conference on the Science of Cancer Health Disparities in Racial/Ethnic Minorities and the Medically Underserved.
Between August 2021 and August 2022, Shannon M. Christy, PhD, assistant member in the Department of Health Outcomes and Behavior at Moffitt Cancer Center in Tampa, Florida, and her colleagues surveyed individuals between the ages of 18 and 26 who were born male and were living in Florida and Puerto Rico, identified as Hispanic or Latino, had sex with a man or were attracted to men, and were able to read and understand Spanish. Among the 102 participants who said they had not received the HPV vaccine, 56% responded incorrectly or “do not know” to a question about whether most sexually active individuals are at risk for being infected with HPV; 20% responded incorrectly or “do not know” to a question assessing whether men can be infected with HPV; and more than half responded incorrectly or “do not know” to questions about the link between HPV and anal (54%), oral (61%), or penile (65%) cancers.
Fifty-six percent did hear about the HPV vaccine, yet only 19% said a provider had recommended it to them. Currently, the Centers for Disease Control and Prevention recommends HPV vaccination for adolescents around ages 11 or 12 and even encourages the vaccination of young adults up to age 26 if they did not receive it when they were younger. The U.S. Food and Drug Administration has approved the HPV vaccine for people ages 9 to 45.

“Sexual minority men are a population group at higher risk for HPV infections and subsequent HPV-related health concerns, including anal cancer,” said Christy. “Prior research has demonstrated suboptimal HPV vaccine uptake among young adults, including sexual minority men. Additional efforts are needed to ensure that all age-eligible community members can benefit from this effective cancer prevention method.”
One place to start would be more Spanish-language materials about HPV vaccination for young adults, as Christy and her collaborators found a lack of such education and no materials culturally adapted for Hispanic and Latino sexual and gender minority community members. “To reduce HPV-related cancer disparities, it is essential that information be relevant and actionable and available to age-eligible people in their preferred language,” said Christy.
Your Genetics Know What You May Not about Cancer Prevention
Whole-exome sequencing can be used as a screening technique to identify if an individual has any genes predisposed for hereditary diseases, including some cancers. The National Comprehensive Cancer Network (NCCN) has established a set of guidelines—including ones for breast, ovarian, and pancreatic cancers and colorectal cancer—to identify individuals who should undergo genetic testing, but guidelines like these might not be catching everyone who should be screened, according to N. Jewel Samadder, MD, a professor of medicine at the Mayo Clinic College of Medicine and co-leader of the precision oncology program at the Mayo Clinic Comprehensive Cancer Center.
“These criteria were created at a time when genetic testing was cost-prohibitive and thus aimed to identify those at the greatest chance of being a mutation carrier in the absence of population-wide whole-exome sequencing,” Samadder said. “However, these conditions are poorly identified in current practice, and many patients are not aware of their cancer risk.”
Samadder presented results from the Tapestry clinical trial at the AACR Annual Meeting 2023 that showed that 39.2% of individuals who consented to whole-exome sequencing and were identified as carriers of predisposition genes for hereditary breast and ovarian cancer (HBOC) or Lynch syndrome would not have qualified under current guidelines. At the time of data cut-off, 44,306 patients from Mayo Clinic sites in Minnesota, Arizona, and Florida had provided a saliva sample. For this part of the trial, researchers used whole-exome sequencing to evaluate samples for BRCA1 and BRCA2, denoting HBOC, and MLH1, MSH2, MSH6, PMS2, and EPCAM, denoting Lynch syndrome.
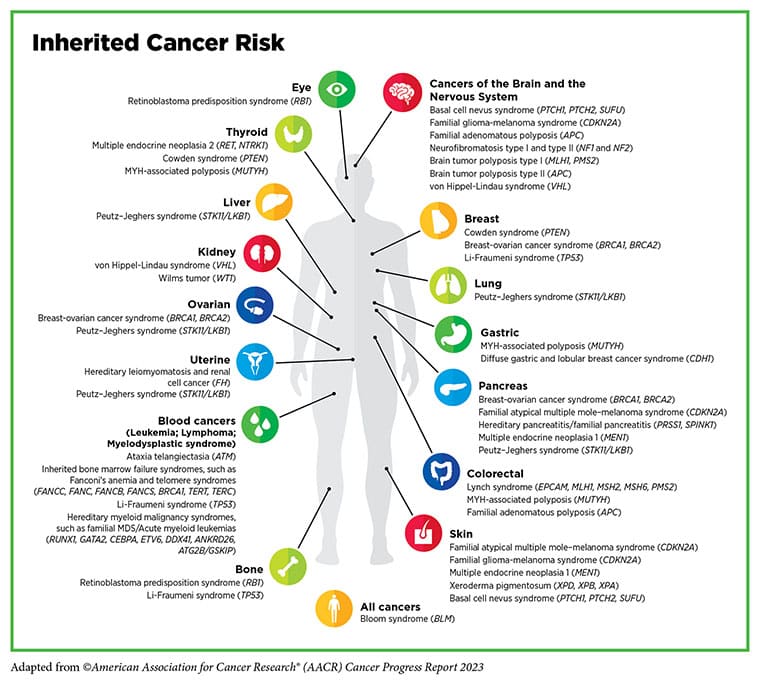
Of the 387 individuals with HBOC and 163 with Lynch syndrome identified, 52.1% did not know prior to this study they had a cancer predisposition condition and 39.2% did not satisfy the existing NCCN criteria for genetic testing. Among the patients who were newly diagnosed with HBOC or Lynch syndrome during this study, 60% were ineligible for genetic testing per the current guidelines. Samadder explained that patients with HBOC have up to an 80% lifetime risk of developing breast cancer and a markedly increased risk, relative to the general population, of developing ovarian cancer, pancreatic cancer, prostate cancer, and melanoma. Meanwhile, patients with Lynch syndrome have up to an 80% lifetime risk of colorectal cancer and up to 60% lifetime risk of endometrial cancer, plus increased risks of upper gastrointestinal, urinary tract, skin, and other cancers.
Knowing about their increased genetic risk can help patients take appropriate next steps, Samadder said. For example, patients with Lynch syndrome can undergo regular colonoscopies, blood and urine screening, and prophylactic hysterectomy, while patients with HBOC can be proactive through advanced breast imaging and prophylactic mastectomy and/or oophorectomy.
“The knowledge that comes from genetics,” Samadder said, “can empower patients to take control of their disease risk and increase their likelihood of avoiding a deadly cancer diagnosis or catching it at an early stage when it is highly curable.”
Like they say, the more you know.
To see how much you know about cancer prevention, take the AACR’s Cancer Prevention Quiz.